Autoimmune Disorders
Autoimmune Disorders Pathology Video
Characterized by tissue damage caused by the immune system.
In the US, the prevalence of autoimmune disorders is 1%.
Autoimmune disorders involve losing tolerance for oneself.
Although self-reactive lymphocytes are regularly produced, they undergo apoptosis (negative selection) in the thymus (T cells) or bone marrow (B cells) or become anergic (because of recognition of antigen in peripheral lymphoid tissues with no 2nd signal).
Autoimmune disorders typically affect women more than men.
Women tend to be in childbearing age.
In people who have a genetic predisposition to autoimmune disorders, the etiology is probably an environmental trigger (increased incidence in twins and associated with certain HLA subtypes).
Key autoimmune disorders to be aware of include:
- Systemic lupus erythematous (SLE)
- Antiphospholipid antibody syndrome
- Sjogren syndrome
- Scleroderma
- CREST syndrome
- Mixed connective tissue disease
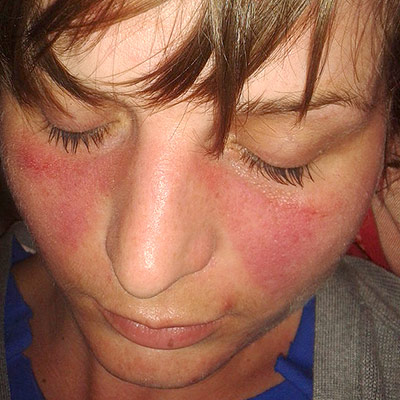
Systemic Lupus Erythematous (SLE)
Systemic lupus erythematous (SLE) which is a systemic autoimmune disease.
Multiple tissues are damaged by antibodies against the host through type I (antigen-antibody complex) and type II (cytotoxic) hypersensitivity.
More typical in females, especially females of African American descent.
Clinical features of systemic lupus erythematous (SLE) include:
- Fever
- Weight loss
- Malar “butterfly” rash, especially after being in the sun
- Arthritis
- Renal damage
- Pleuritis and pericarditis (involvement of serosal surfaces)
- CNS psychosis
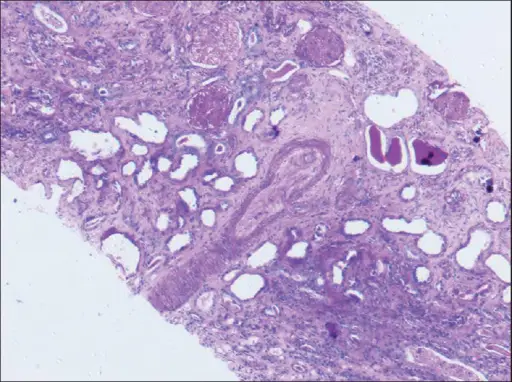
The most frequent type of renal damage is diffuse proliferative glomerulonephritis, while other injury patterns can also occur.
Endocarditis, myocarditis, or pericarditis (may affect any layer of the heart).
Non-infectious (sterile) vegetations on the heart valves may also be present on both sides of the valve, causing Libman-Sacks endocarditis.
Leukopenia, thrombocytopenia, or anemia (because of autoantibodies against cell surface proteins).
Systemic lupus erythematous (SLE) lab values include:
- Antinuclear antibody (sensitive, but non-specific)
- Anti-double stranded DNA antibodies (highly specific)
Drug-induced systemic lupus erythematous (SLE) is characterized by antihistone antibodies.
Common causes of drug induced systemic lupus erythematous (SLE) include hydralazine, procainamide, and isoniazid.
Remission of drug induced systemic lupus erythematous (SLE) frequently follows drug withdrawal.
Infection and renal failure are the usual causes of death in systemic lupus erythematous (SLE).
Antiphospholipid Syndrome
Antiphospholipid syndrome and systemic lupus erythematous (SLE) are related (30 percent of cases).
Characterized by the presence of autoantibodies against phospholipid-bound proteins.
The two most prevalent antibodies are:
- Anticardiolipin antibody (which causes false positive syphilis tests)
- Lupus anticoagulants (which falsely elevates PTT lab values)
Antiphospholipid syndrome leads to arterial and venous thrombosis including:
- Deep venous thrombosis
- Hepatic vein thrombosis
- Placental thrombosis (repeated pregnancy loss)
- Stroke
Lifelong anticoagulation is needed.
Sjögren’s Syndrome
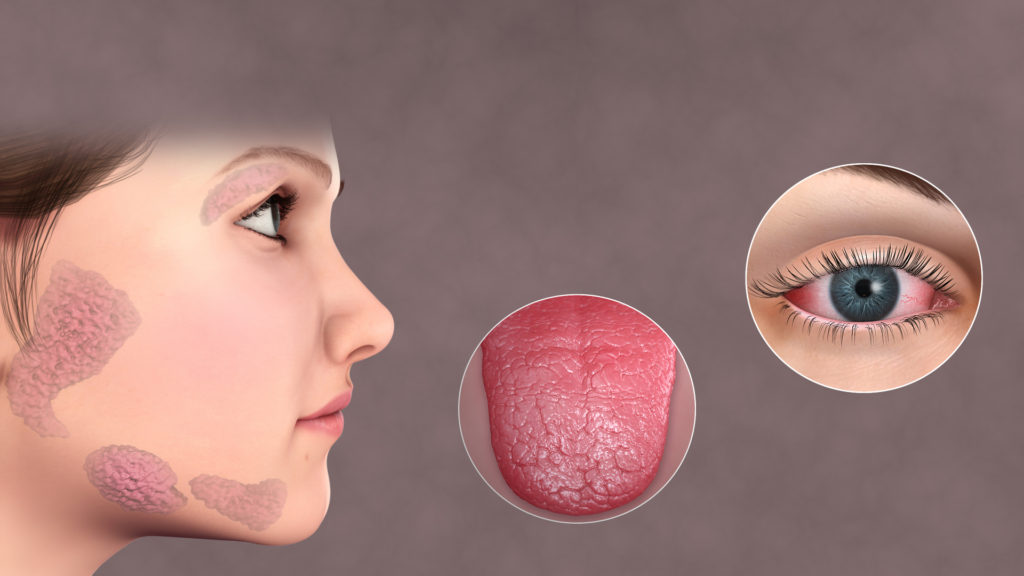
Sjögren’s syndrome is the autoimmune destruction of lacrimal and salivary glands.
Fibrosis coupled with lymphocyte-mediated injury (type IV hypersensitivity reaction).
In an older lady, dry eyes (keratoconjunctivitis), dry mouth (xerostomia), and recurring dental caries are the typical symptoms (50-60 years)
Symptoms of Sjögren’s syndrome typically include:
- Dry eyes
- Dry mouth
- Trouble swallowing
ANA and anti-ribonucleoprotein antibodies (anti-SS-A/Ro and anti-SS-B/La) are the defining features.
ANA and anti-ribonucleoprotein antibodies (anti-SS-A/Ro and anti-SS-B/La) are the defining features.
Frequently linked to other autoimmune diseases, especially rheumatoid arthritis.
An increased chance of developing B-cell (marginal zone) lymphoma, which late in the course of the disease manifests as unilateral enlargement of the parotid gland.
Scleroderma
Autoimmune tissue injury accompanied by fibroblast activation and collagen deposition (fibrosis).
Categorized into diffuse and localized types.
The skin and early visceral involvement are signs of the diffuse kind.
Almost any organ can be damaged; the esophagus is frequently impacted, leading to irregular motility (dysphagia for solids and liquids).
ANA and anti-DNA topoisomerase I (Scl-70) antibody are the defining features.
Local skin and late visceral involvement are exhibited in the local type.
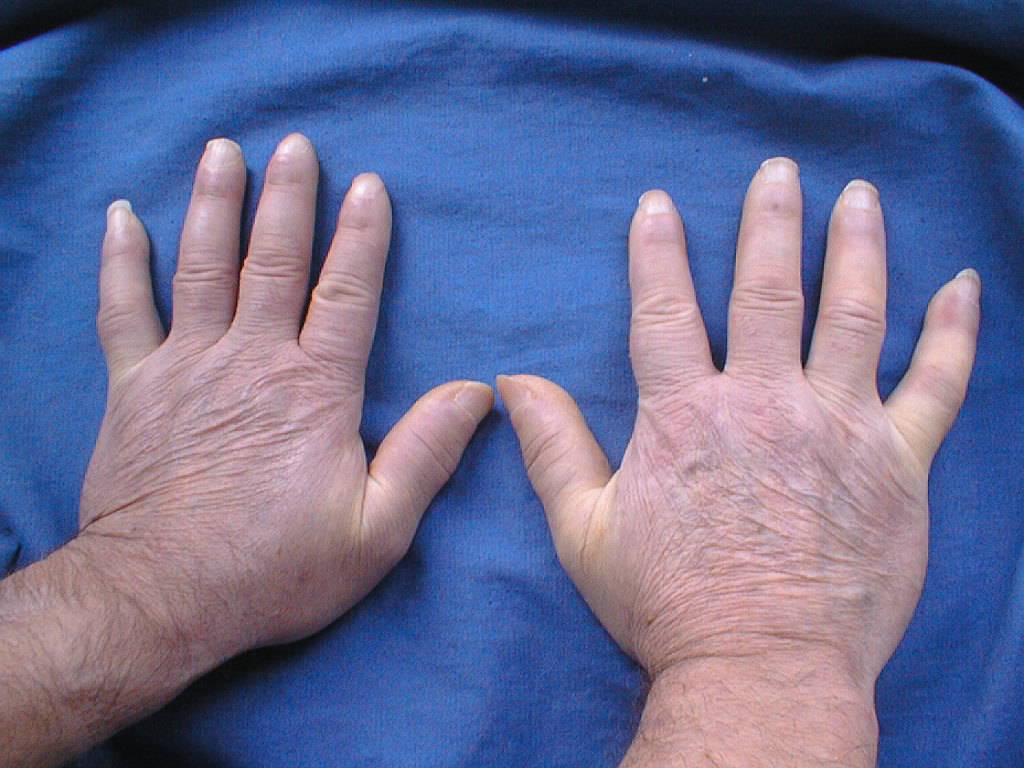
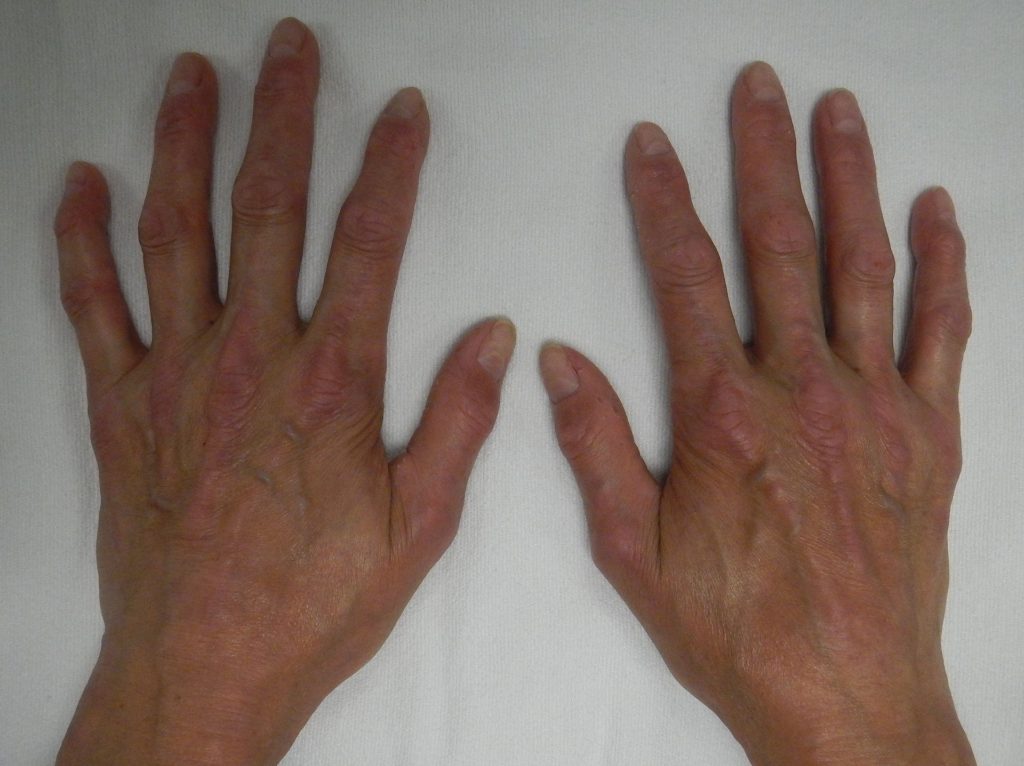
CREST Syndrome
The CREST syndrome involves:
- Calcinosis cutis
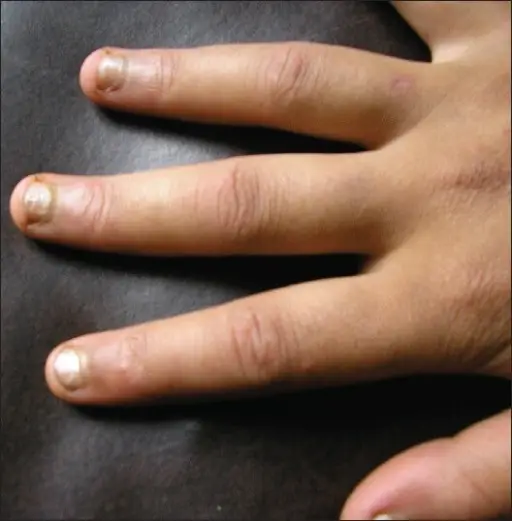
- Raynaud syndrome
- Esophageal dysmotility
- Sclerodactyly
- Telangiectasias
Mixed Connective Tissue Disease
Mixed connective tissue disease is an autoimmune-mediated tissue damage with mixed characteristics of SLE, systemic sclerosis, and polymyositis.
Serum antibodies against Ul ribonucleoprotein are the defining features.