Thrombosis
Thrombosis and Embolism Pathology Video
Pathologic development of an intravascular blood clot may result in a thrombus.
Thrombosis may occur in arteries and/or veins.
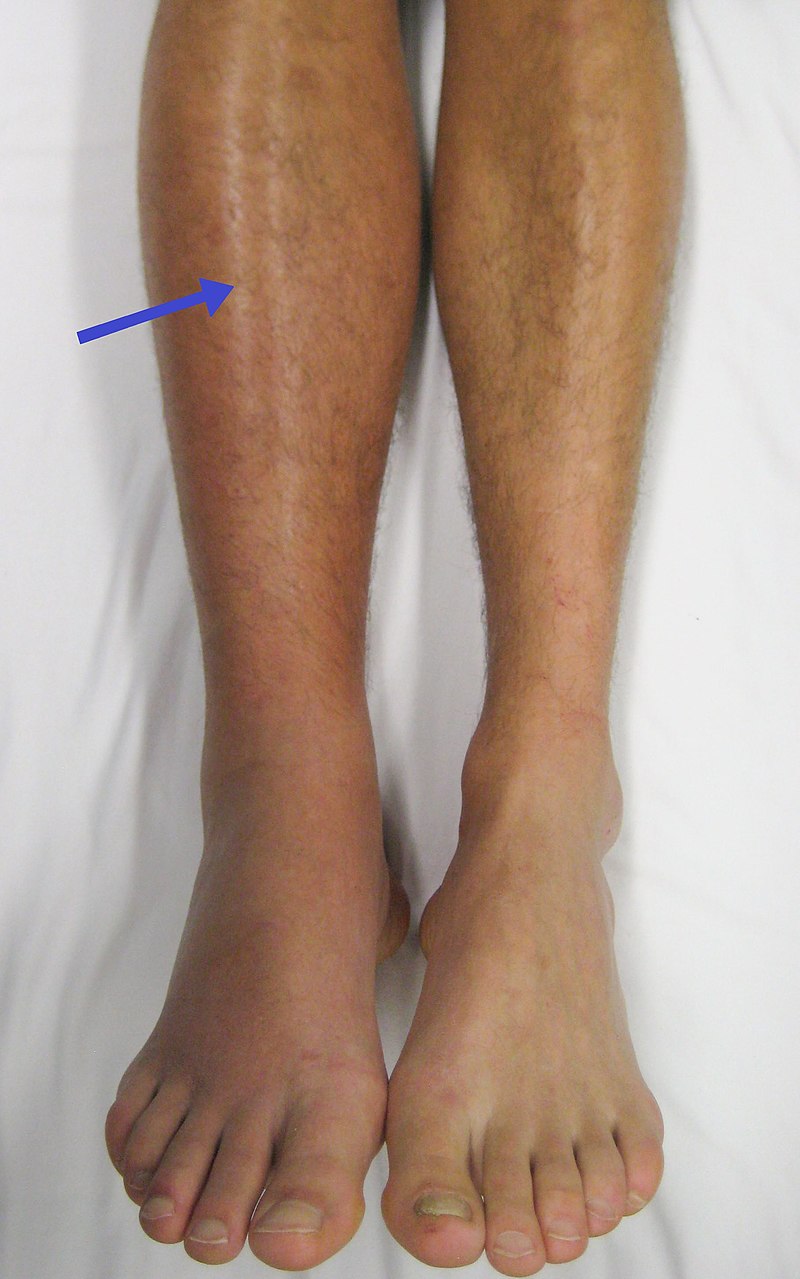
The deep veins in the leg below the knee are where they are most frequently found.
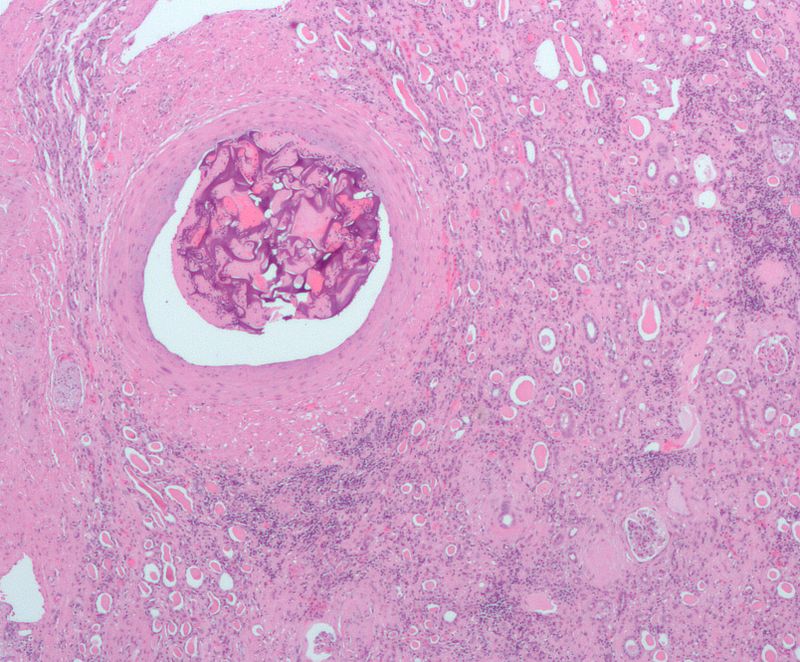
Ante-mortem thrombosis is characterized by lines of Zahn and adhesion to vessel walls.
Lines of Zahn are alternating layers of platelets/fibrin and red blood cells (RBCs).

Note that postmortem clots are not attached to the vessel wall and do not have lines of Zahn.
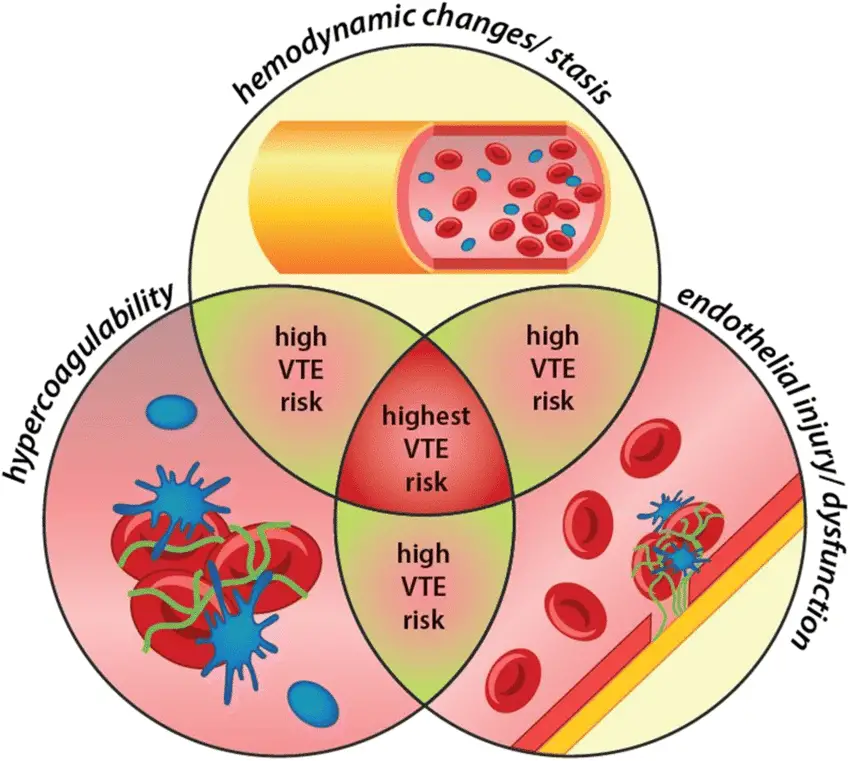
Blood flow disturbance, endothelial cell injury, and a hypercoagulable condition are three key risk factors for thrombosis which are described as Virchow’s triad.
Disruption in Normal Blood Flow
Blood flow turbulence and stasis raise the risk of thrombosis.
Laminar blood flow, which is typical, keeps platelets and other components scattered and inactive.
Potential causes of disruption in normal blood flow include:
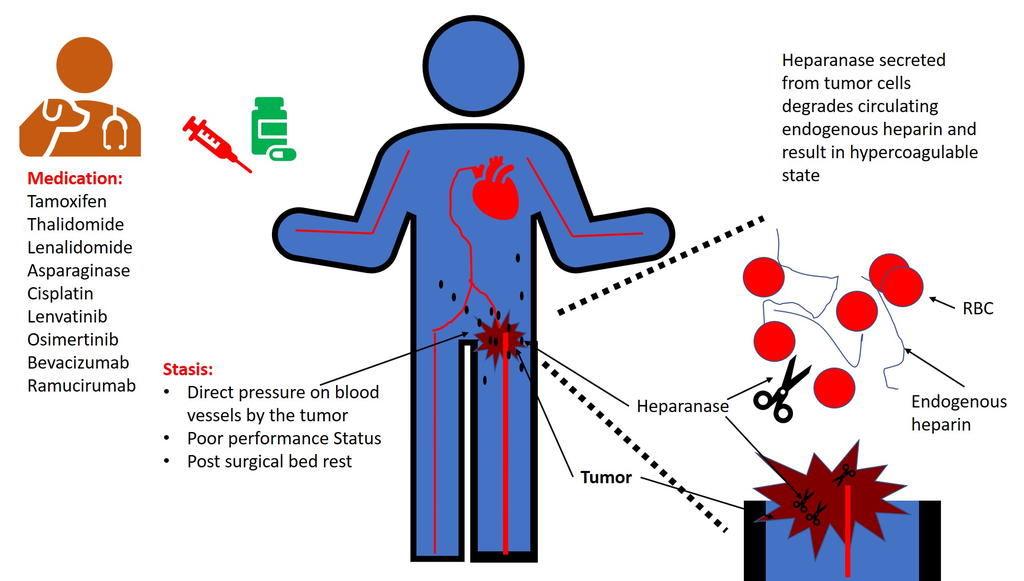
- Immobilization which increases risk of deep venous thrombosis
- Cardiac wall dysfunction such as arrhythmia or myocardial infarction
- Aneurysm
Endothelial Cell Damage
Endothelial damage disrupts endothelial cells’ protective role, raising the possibility of thrombosis.
Multiple mechanisms are used by endothelial cells to prevent thrombosis which includes:
- Reduce exposure to underlying tissue factor and sub-endothelial collagen
- Creates prostacyclin (PGI 2)
- NO production
- Platelet aggregation inhibition
- Release compounds similar to heparin that boost the antithrombin lU (ATI) enzyme, which renders thrombin and coagulation factors inactive
- Secrete tissue plasminogen activator (TPA) which changes plasminogen to plasmin, cleaves fibrin and serum fibrinogen, destroys coagulation factors, and blocks platelet aggregation
- Secrete thrombomodulin which causes thrombin to be redirected to activate protein C, deactivating factors V and VI
Damage to endothelial cells are brought on by vasculitis, atherosclerosis, and elevated homocysteine levels.
Low levels of folate and vitamin B12 raise homocysteine, which raises the risk of thrombosis.
Tetrahydrofolate, or folic acid, circulates in the blood as methyl-THF.
Cobalamin (vitamin Bl2) receives a methyl transfer, enabling THF to take part in the production of DNA precursors.
Methyl is added to homocysteine by cobalamin, which produces methionine.
Homocysteine builds up because the conversion of homocysteine to methionine is reduced when folate or vitamin B12 deficiency is present.
Cystathionine Beta Synthase Deficiency
Homocysteine levels are high and homocystinuria occurs as a result of cystathionine beta synthase (CBS) deficiency.
Homocysteine is converted by cystathionine beta synthase (CBS) to cystathionine.
A lack of the enzyme cystathionine beta synthase (CBS) causes an accumulation of homocysteine.
Hypercoagulable State
A hypercoagulable state may be due to an excess of procoagulant proteins or defective anticoagulant proteins.
Hypercoagulable states may be inherited or acquired.
Recurrent deep vein thrombosis (DVT) in children is an example of the classic presentation.
Usually happens in the deep leg veins, but it can also happen in the hepatic and cerebral veins.
The autosomal dominant protein C or S deficiency reduces the coagulation cascade’s negative feedback.
Factors V and VII are often deactivated by proteins C and S.
Because of their shorter half-lives compared to factors II, VII, IX, and X; proteins C and S experience a transitory shortfall during the first stage of warfarin therapy.
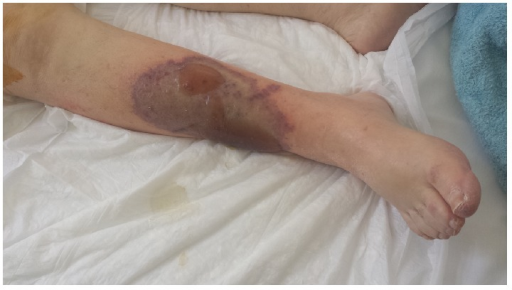
When warfarin therapy is started in patients with preexisting C or S deficit, a significant deficiency is present, increasing the risk of thrombosis, particularly in the skin-causing warfarin skin necrosis.
A mutant of factor V called factor V Leiden lacks the cleavage site needed for proteins C and S to deactivate it.
The most frequent hereditary factor for hypercoagulability.
An hereditary point mutation in prothrombin called prothrombin 202lOA leads to enhanced gene expression.
Increased prothrombin causes a rise in thrombin, which encourages the formation of thrombus.
Lack of ATIll increases the risk of thrombus by reducing the protective action of heparin-like molecules generated by the endothelium.
Heparin-like substances typically cause ATI to be activated, which renders thrombin and coagulation factors inactive.
In ATIll deficiency, PTT doesn’t increase with standard heparin dosing.
Pharmacologic heparin functions by interacting with and turning on ATlll.
Coumadin is then administered to maintain an anticoagulated condition after high heparin dosages activate minimal ATI.
A hypercoagulable state is linked to oral contraceptives.
The increased generation of coagulation factors caused by estrogen raises the danger of thrombosis.
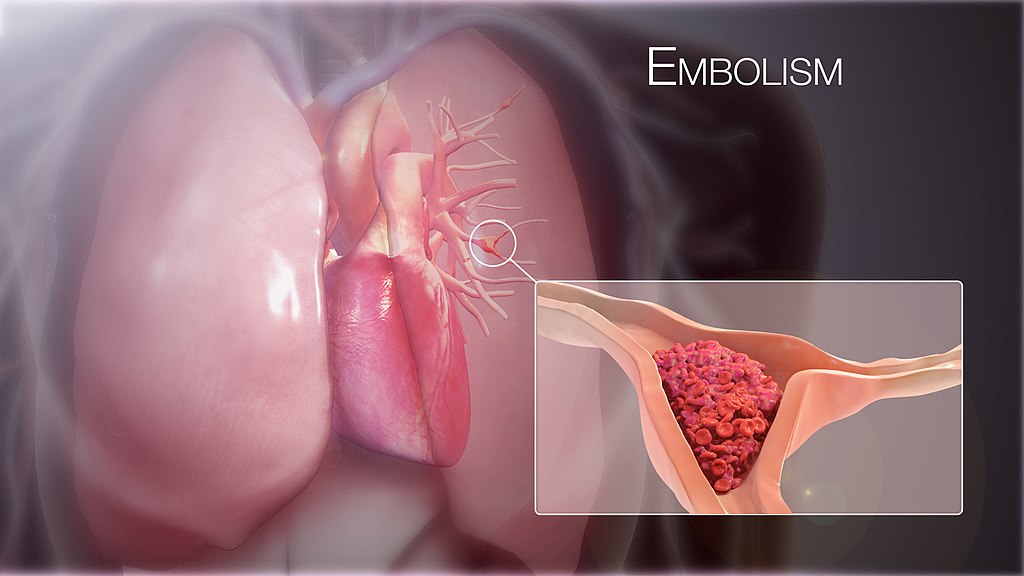
Embolism
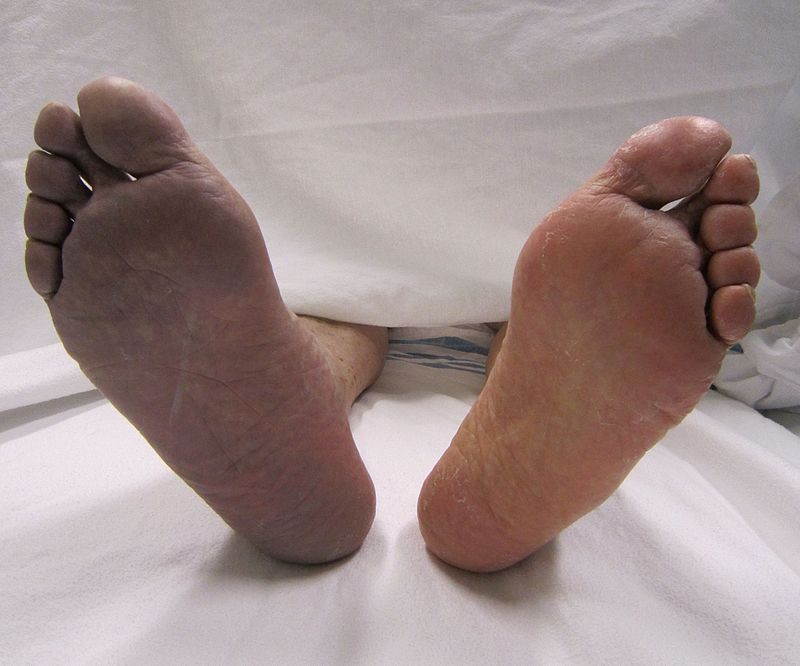
An embolism is an intravascular mass that moves and obstructs downstream vessels.
Embolism symptoms depend on the vessel involved.
A thrombus that dislodges is the cause of thromboembolism.
Types of emboli include:
- Thromboembolism (most common)
- Atherosclerotic embolism (will have cholesterol clefts
- Fat embolism (will contain adipose tissue)
- Gas embolism (divers)
- Amniotic fluid embolism (pregnant lady giving birth)
Thromboembolism
With a prevalence of more than 95%, thromboembolism is regarded as the most prevalent kind of embolus.
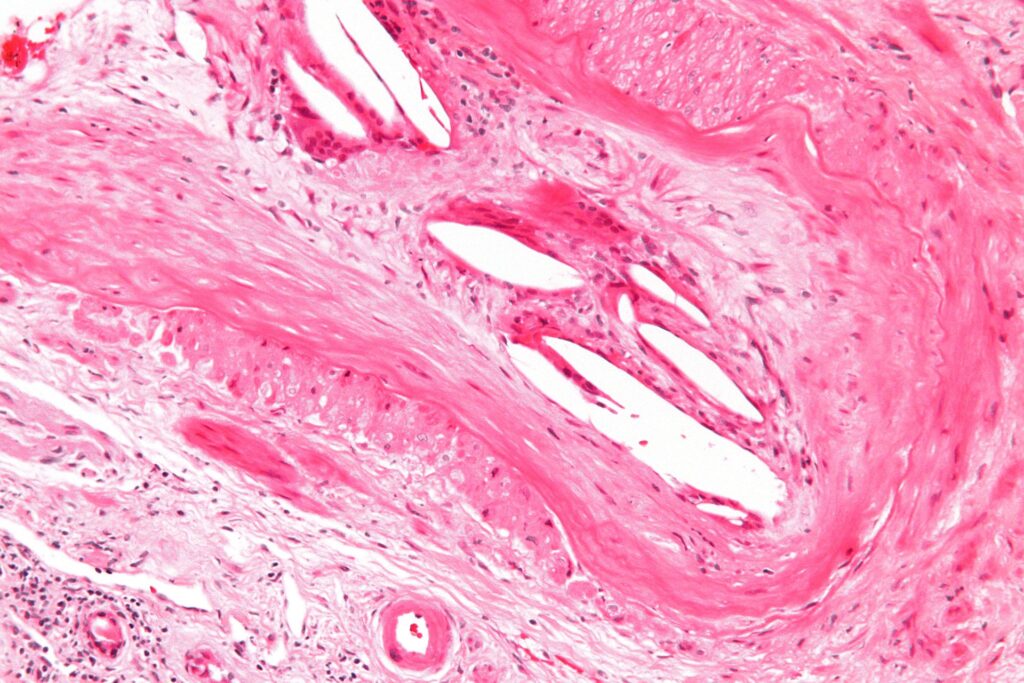
Atherosclerotic embolus
A dislodged atherosclerotic plaque is the cause of an atherosclerotic embolus.
The presence of cholesterol clefts in the embolus characterizes atherosclerotic embolus.
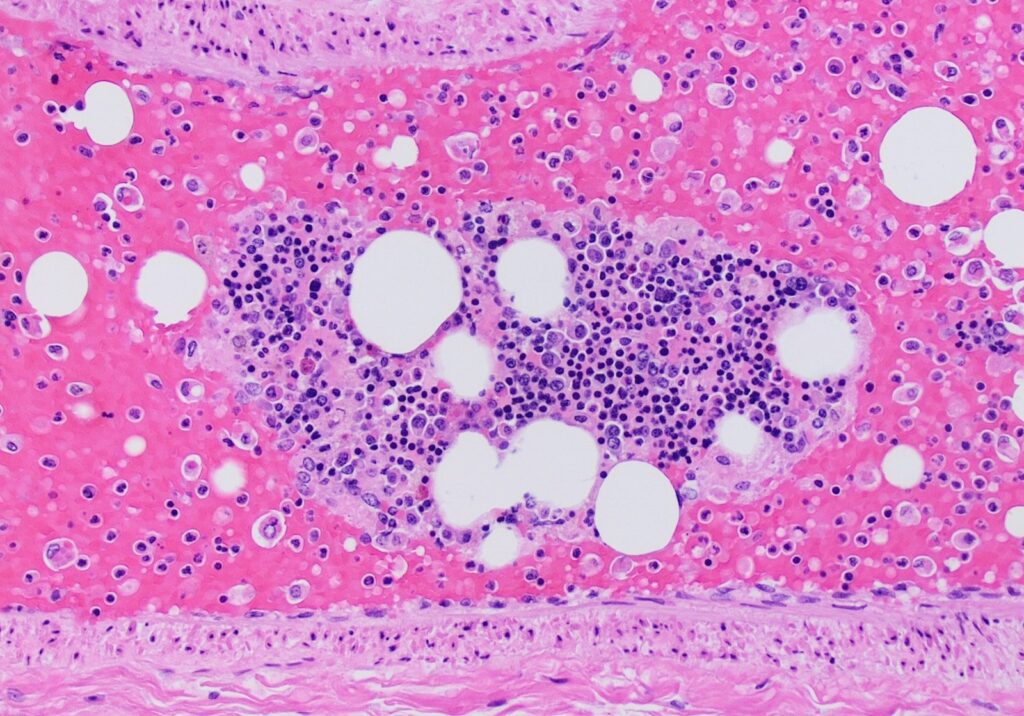
Fat emboli
Bone fractures, especially lengthy bone fractures, and soft tissue injuries are linked to fat embolism.
Fat emboli appears while there is still a fracture or soon after repair.
Fat emboli are characterized by dyspnea (fat, often with bone marrow elements, is seen in pulmonary vessels) and petechiae on the skin overlying the chest.
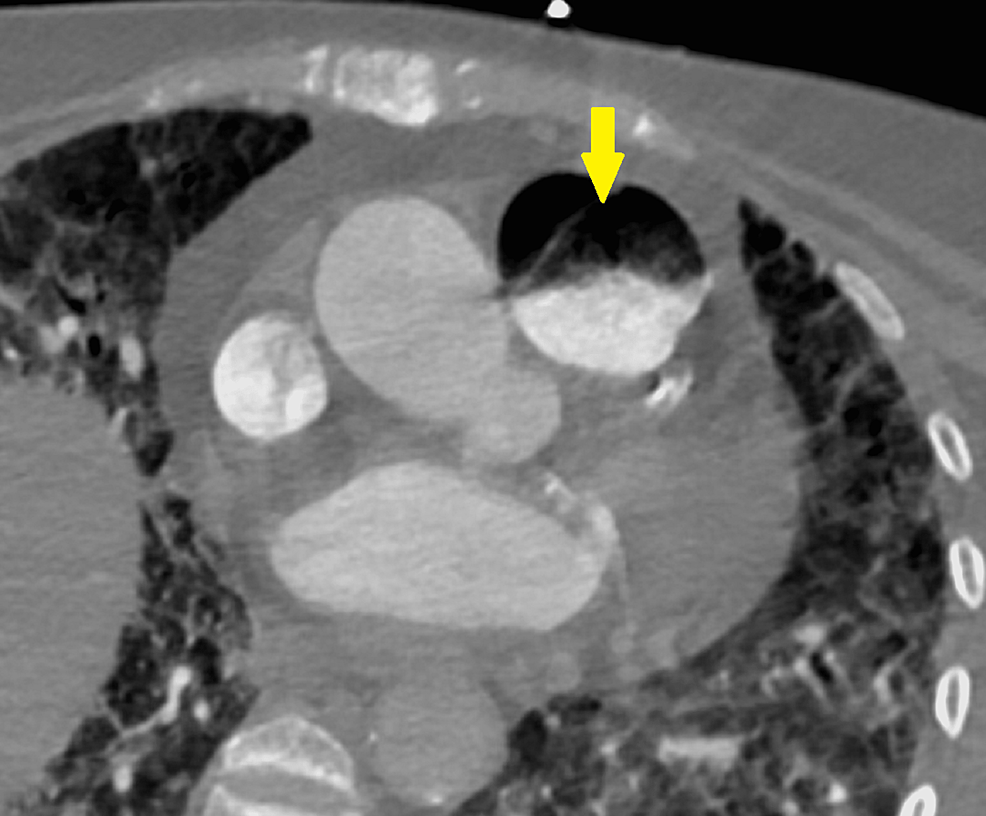
Gas embolism
Gas embolism is classically seen in decompression sickness.
A diver’s quick ascent causes nitrogen gas to precipitate out of the diver’s blood.
Gas embolism presents with joint and muscle pain (‘bends’) and respiratory symptoms (‘chokes’).
Multifocal ischemic bone necrosis is a characteristic of the chronic form of the gas embolism known as Caisson disease.
Additionally, when air is pushed into the abdomen during laparoscopic surgery, gas emboli can happen.
Amniotic embolism
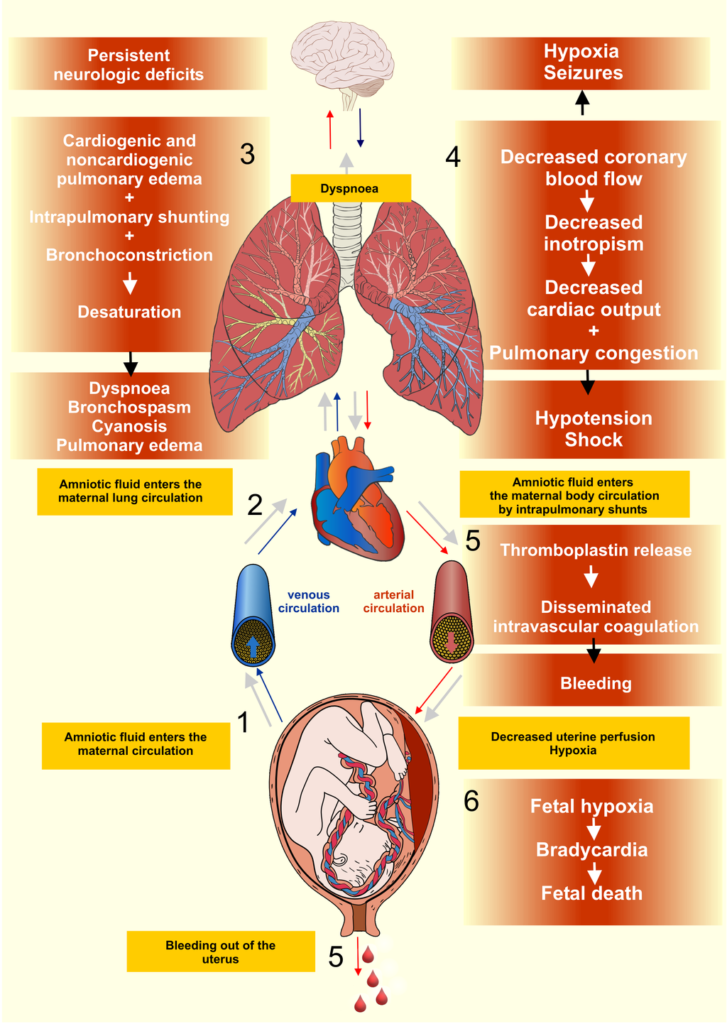
During labor or delivery, amniotic fluid embolism enters the maternal bloodstream.
Shortness of breath, neurological symptoms, and disseminated intravascular coagulation (DIC) are the symptoms of this illness, which is brought on by the thrombogenic nature of amniotic fluid.
Amniotic emboli are characterized by squamous cells and keratin debris, from fetal skin, in the embolus.
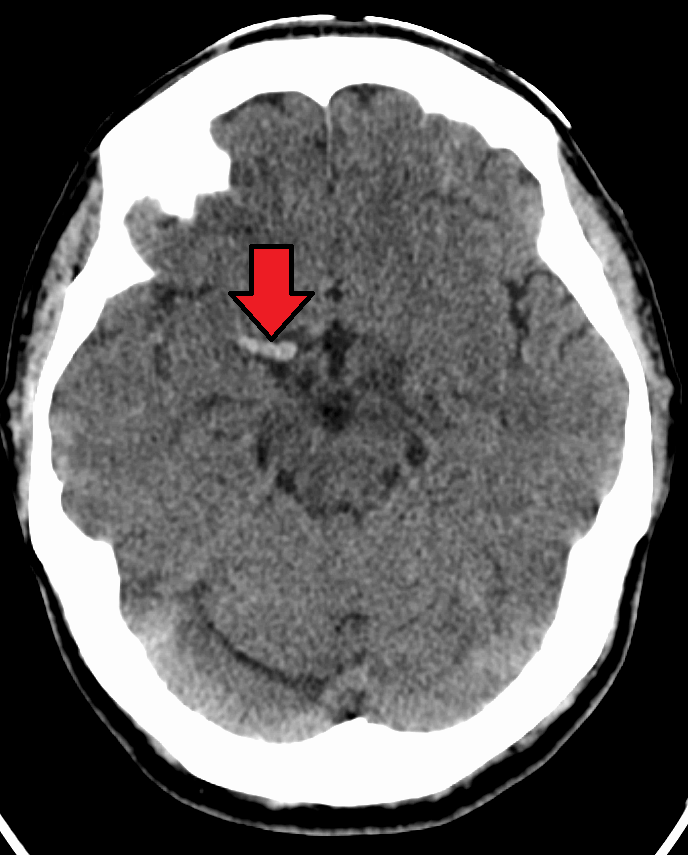
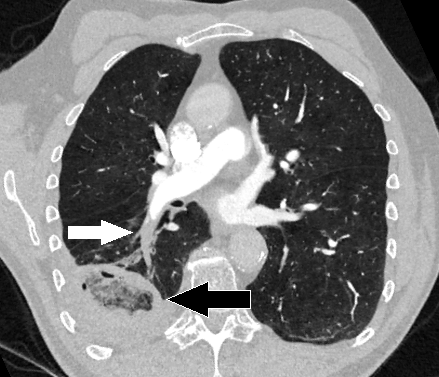
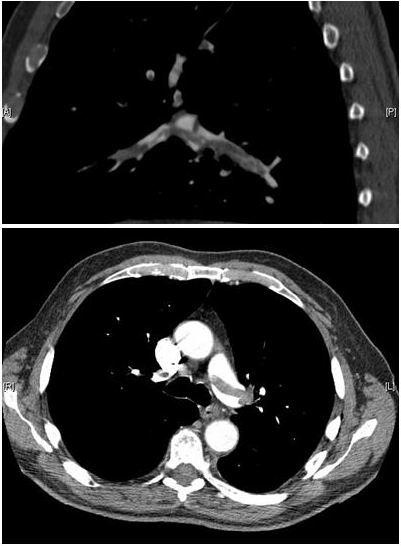
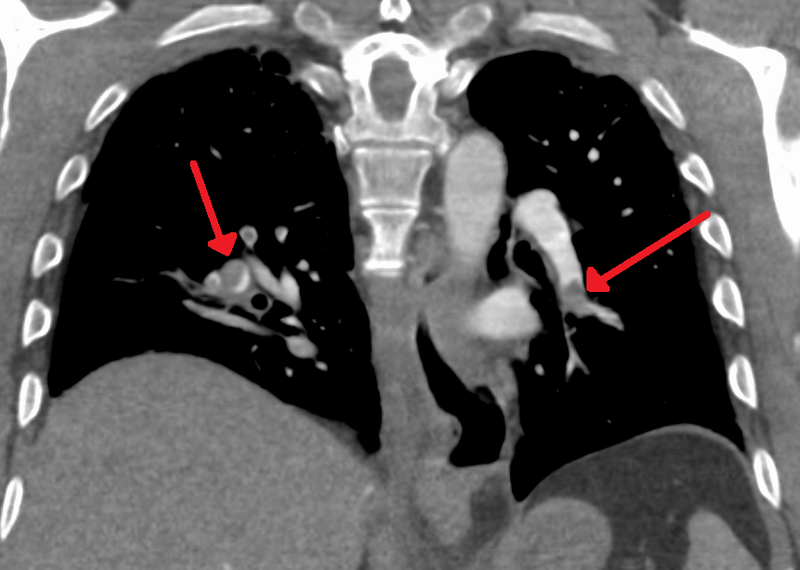
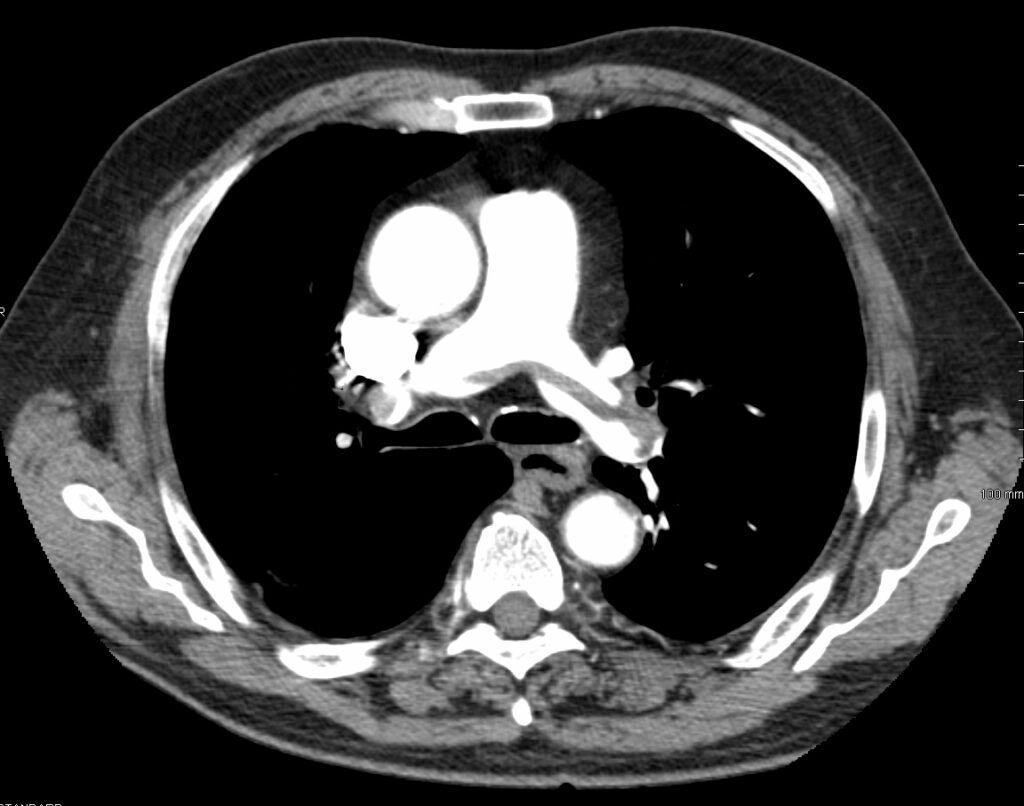
Pulmonary Embolism (PE)
The main cause of pulmonary embolism (PE) is thromboembolism.
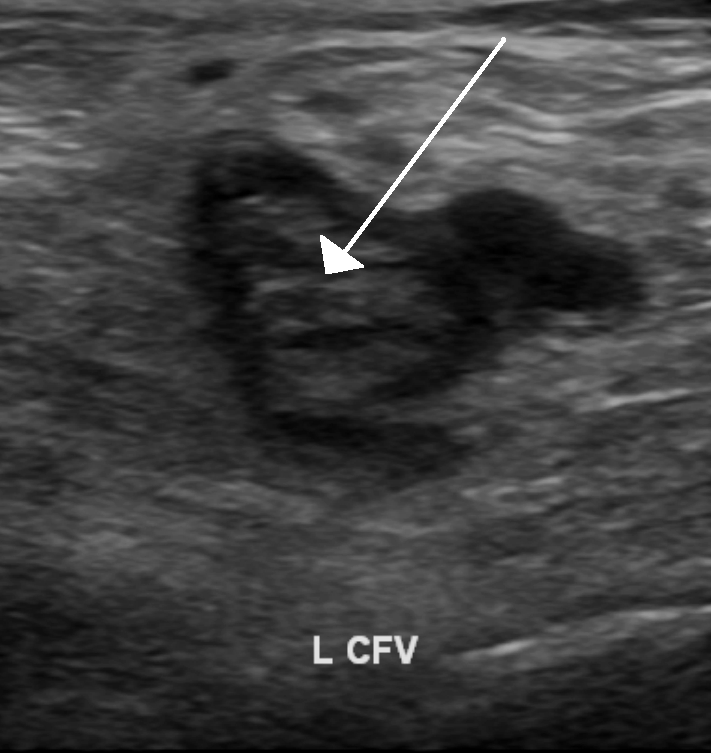
Deep venous thrombosis (DVT) in the lower extremities, typically involving the femoral, iliac, or popliteal veins, is the most frequent cause.
Because the lung receives blood from both the pulmonary and bronchial arteries, it is frequently clinically quiet.
The embolus typically shrinks on its own and is modest.
Only 10% of pulmonary embolisms (PEs) result in pulmonary infarction, which happens when a large or medium-sized artery is obstructed in individuals with pre-existing cardiac impairment.
The symptoms of pulmonary embolism (PE) includes:
- Shortness of breath
- Hemoptysis
- Pleuritic chest pain
- Pleural effusion
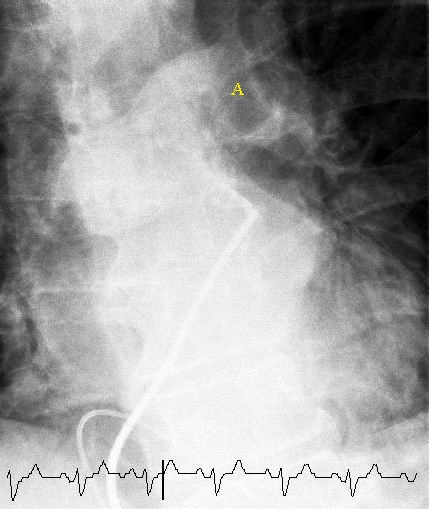
V/Q lung scan, short for lung or pulmonary ventilation (V) and perfusion (Q), shows mismatch and perfusion is abnormal.
On the other hand, a vascular filling defect in the lung is seen on spiral CT.
Doppler ultrasonography on the lower extremities can help identify deep vein thrombosis (DVT).
D-dimer is elevated in pulmonary embolism (PE).
Autopsy examination reveals a hemorrhagic, wedge-shaped infarct.
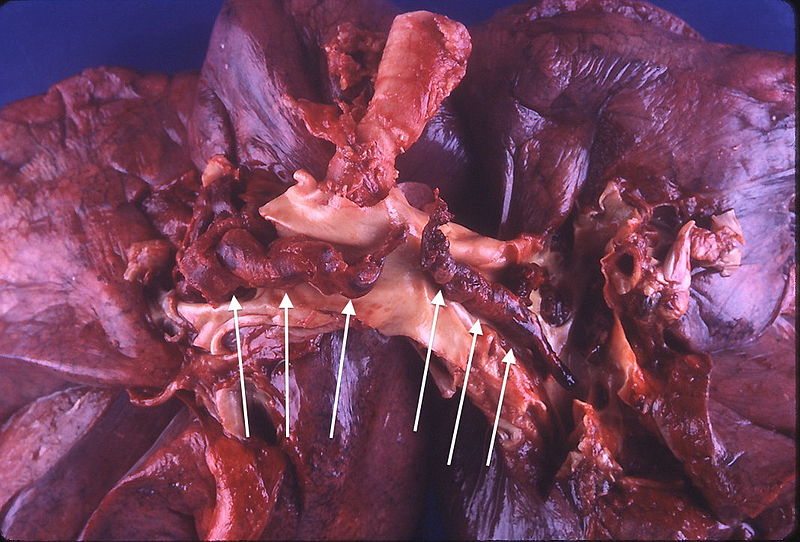
When both the left and right pulmonary arteries are significantly occluded by a massive saddle embolus, or when a large pulmonary artery is suddenly blocked, sudden mortality results.
An electromechanical dissociation causes death.
Chronic emboli that rearrange over time may cause pulmonary hypertension.
Systemic Embolism
Thromboembolism is typically the cause of systemic embolism, which most frequently develops in the left heart.
It is a critical side effect of classical orthotopic heart transplantation.
Systemic emboli travels through the systemic circulation to obstruct blood flow to organs, most frequently the lower limbs.