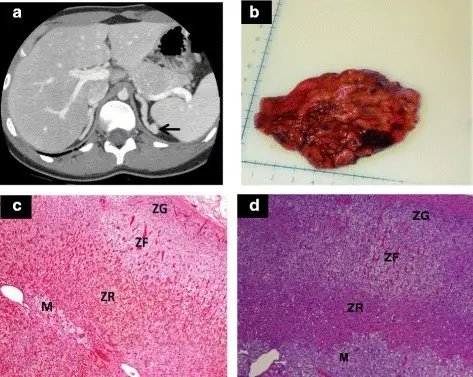
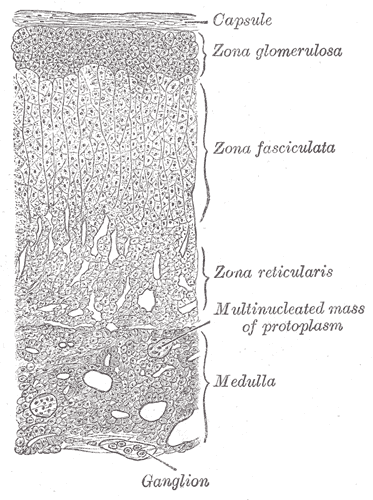
Adrenal Cortex
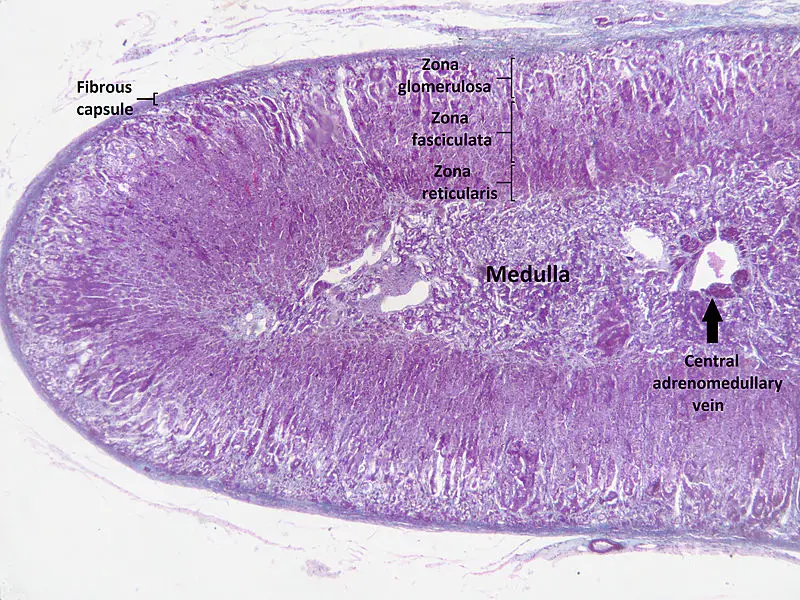
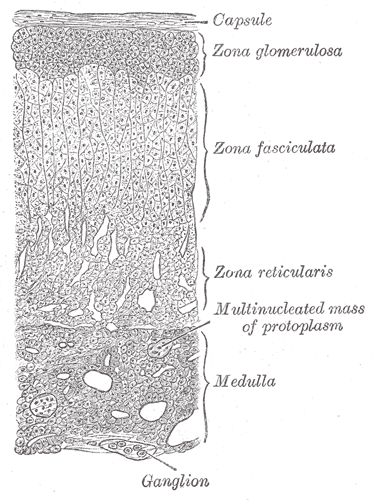
The largest and outermost portion of an adrenal gland is called the adrenal cortex.
The adrenal gland is composed of three distinct zones which include:
- Zona glomerulosa
- Zona fasciculata
- Zona reticularis
Zona glomerulosa produces mineralocorticoids like aldosterone.
Zona fasciculata creates glucocorticoids like cortisol.
Zona reticularis generates sex hormones like testosterone.
Pathology of the adrenal gland includes:
- Cushing syndrome (hypercortisolism)
- Conn syndrome (hyperaldosteronism)
- Congenital adrenal hyperplasia
- Adrenal insufficiency
- Adrenal medulla
- Pheochromocytoma
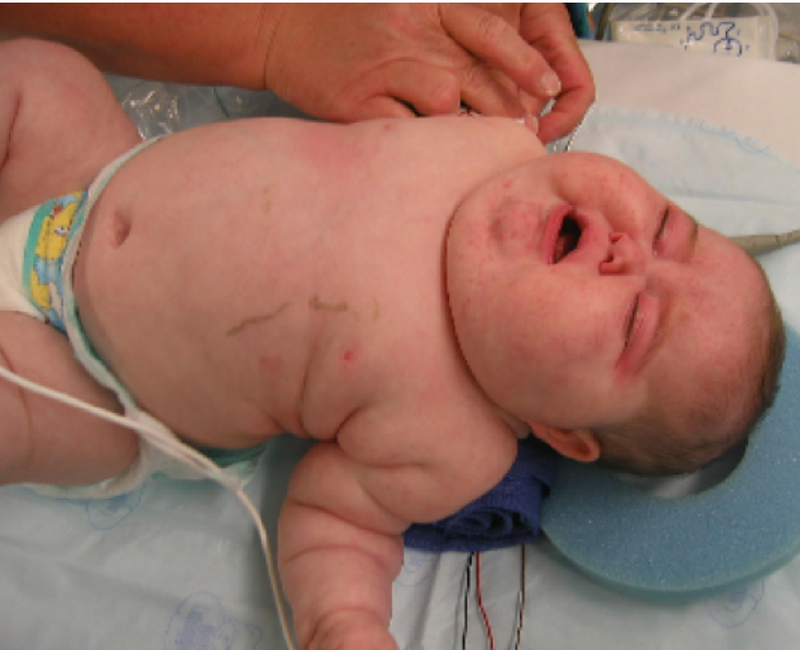
Cushing Syndrome (Hypercortisolism)
When there is too much cortisol in the body, Cushing’s syndrome develops.
The stress hormone or cortisol, is essential for controlling blood sugar and converting food into energy.
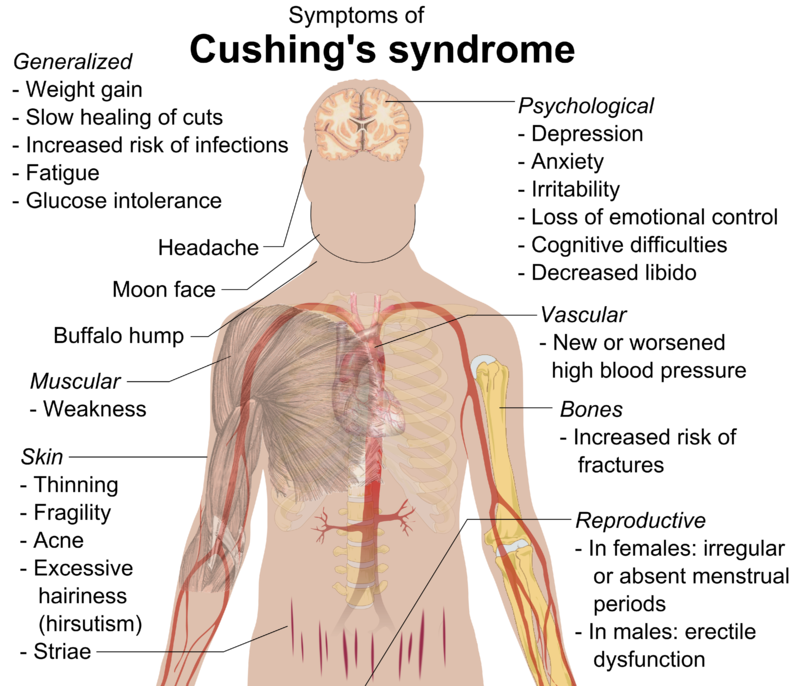
There are several clinical features of Cushing syndrome includes:
- Muscle weakness
- Thin extremities when cortisol breaks down muscle for gluconeogenesis
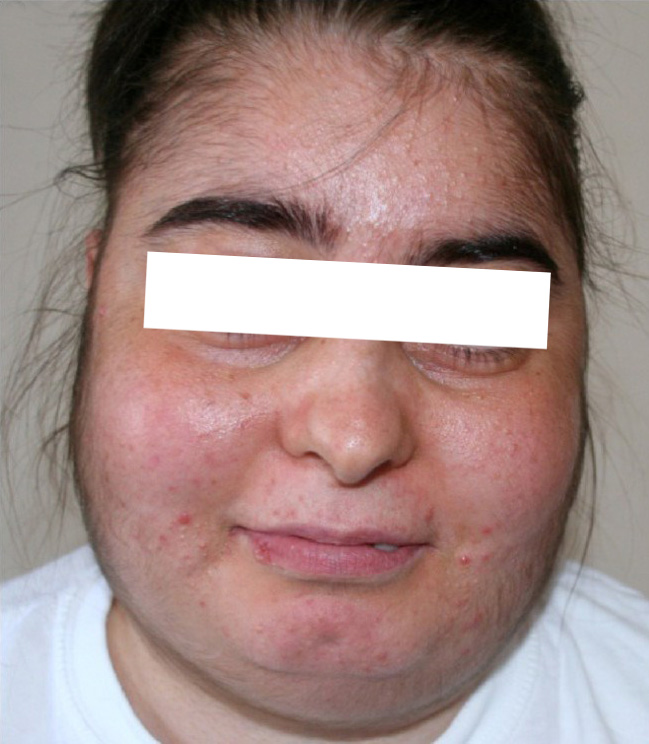
- Moon facies
- Buffalo hump
- Truncal obesity with high insulin due to high glucose increased storage of fat
- Hypertension
- Osteoporosis
- Immunosuppression
- Abdominal striae due to impaired collagen synthesis with skin thinning
Diagnosis of Cushing syndrome is made by checking for increased 24-hour urine cortisol levels.
Causes of Cushing syndrome include:
- Exogenous corticosteroids because they produce bilateral adrenal atrophy
- Steroids reduce the release of adrenocorticotropic hormone (ACTH), which results in a negative feedback
- Primary adrenal adenoma
- Hyperplasia, or cancer are other causes that result in atrophy of the unaffected adrenal gland
- Pituitary adenoma that secretes adrenocorticotropic hormone (ACTH)
- Paraneoplastic adrenocorticotropic hormone (ACTH) secretion from diseases like small cell lung cancer both cause bilateral adrenal hyperplasia
Dexamethasone (a cortisol analog) at high doses prevents a pituitary adenoma from producing adrenocorticotropic hormone (ACTH), which lowers cortisol levels.
Cortisol levels continue to be elevated because dexamethasone does not stop a small cell lung cancer from producing ectopic adrenocorticotropic hormone (ACTH).
Conn Syndrome (Hyperaldosteronism)
Conn syndrome is a rare disorder known as primary aldosteronism.
Conn syndrome is brought on by an excess of the hormone aldosterone, which regulates the levels of sodium and potassium in the blood.
Conn syndrome presents as:
- Hypertension
- Metabolic alkalosis
- Hypokalemia
- Hypernatremia
In the distal tubules and collecting duct, aldosterone enhances potassium and hydrogen ion secretion and sodium absorption.
Increased absorption of sodium expands plasma volume leading to hypertension.
Adrenal adenomas are the most frequent cause of Conn syndrome, but sporadic adrenal hyperplasia and adrenal hyperplasia are less frequent causes.
Conn syndrome is characterized by high aldosterone and low renin where high blood pressure down regulates renin via negative feedback.
On the other hand, renin-angiotensin system activation such as renovascular hypertension or congestive heart failure (CHF) is associated with secondary hyperaldosteronism.
High levels of renin and aldosterone are defining characteristics of Conn syndrome.
Congenital Adrenal Hyperplasia (CAH)
A group of hereditary diseases affecting the adrenal glands, a pair of walnut-sized structures above the kidneys, is known as congenital adrenal hyperplasia (CAH).
Congenital adrenal hyperplasia (CAH) is due to the excess sex steroids with hyperplasia of both adrenal glands.
The most common cause of congenital adrenal hyperplasia is inherited 21-hydroxylase deficiency.
Aldosterone and corticosteroids are made possible by the enzyme 21-hydroxylase.
When an enzyme is lacking, steroidogenesis is primarily diverted toward the creation of sex steroid, which does not require 21-hydroxylase.
Lack of negative feedback from increased adrenocorticotropic hormone (ACTH) secretion brought on by cortisol deficiency causes bilateral adrenal hyperplasia.
Clinical features of congenital adrenal hyperplasia (CAH) include:
- Hyperkalemia
- Salt wasting with hyponatremia
- Hypovolemia due to lack of aldosterone
- Clitoral enlargement in women
- Precocious puberty in men
- Hypotension
Adrenal Insufficiency
Adrenal insufficiency, commonly known as Addison’s disease, is a rare disorder that develops when the body doesn’t generate enough of specific hormones.
Waterhouse-Friderichsen syndrome, a group of symptoms caused when the adrenal glands fail to function normally that can cause acute insufficiency.
Features of Waterhouse-Friderichsen syndrome include:
- Hemorrhagic necrosis of the adrenal glands, which is typically brought on by disseminated intravascular coagulation (DIC) in young children who have Neisseria meningitidis infection
- Hypotension is made worse by cortisol deficiency, frequently resulting in death
Chronic adrenal insufficiency, or Addison disease as it is commonly known, is brought on by the gradual loss of the adrenal glands.
Common causes of Waterhouse-Friderichsen syndrome include:
- Autoimmune destruction which is the most common cause in the West
- Tuberculosis is also a common cause in the developing world
- Metastatic carcinoma such as cancers arising from lung
Symptoms of Waterhouse-Friderichsen syndrome include:
- Hypotension
- Hyponatremia
- Hypovolemia
- Hyperkalemia
- Weakness
- Vomiting
- Diarrhea
- Hyperpigmentation
Hyperpigmentation is due to proopiomelanocortin (POMC) increased adrenocorticotropic hormone (ACTH) byproducts stimulating melanocytic pigment production.
Adrenal Medulla
The adrenal medulla is the innermost part of your adrenal gland.
Adrenal medulla hormones include adrenaline and noradrenaline.
The adrenal medulla is the primary physiological source of catecholamines and are made up of neural crest-derived chromaffin cells.
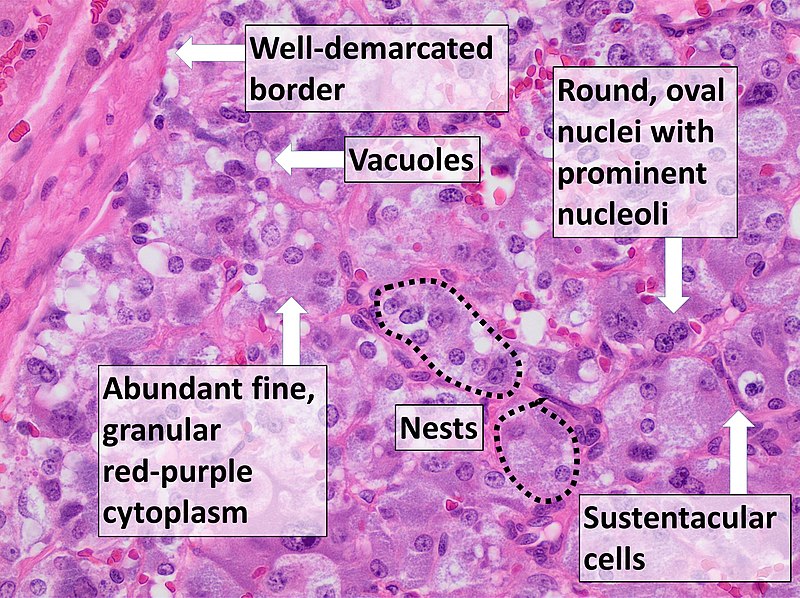
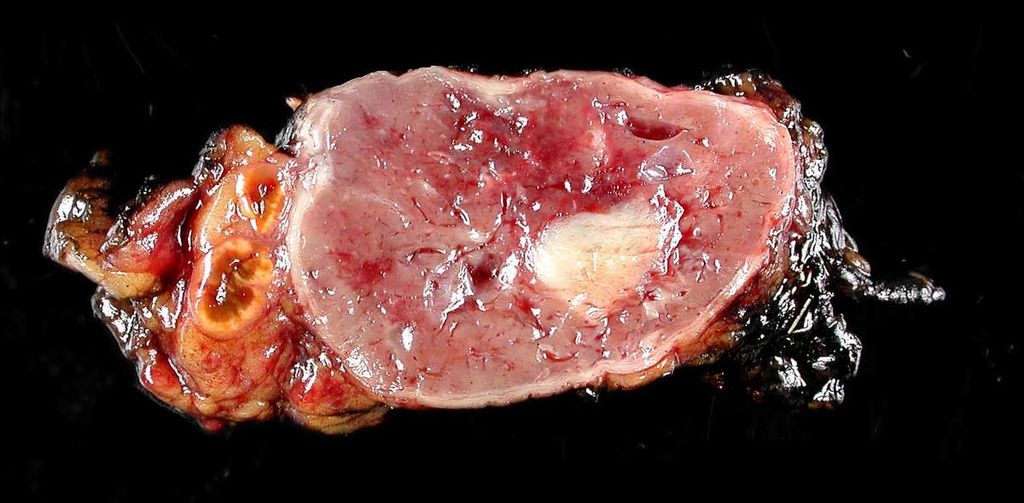
Pheochromocytoma
A particular kind of neuroendocrine tumor (NET) known as a pheochromocytoma develops from chromaffin cells.
Chromaffin cells are located in the adrenal glands and create hormones that the body requires.
Clinical features of pheochromocytoma results from elevated serum catecholamine levels.
Clinical features of pheochromocytoma include:
- Perspiration
- Tachycardia
- Palpitations
- Headaches
- Episodic hypertension
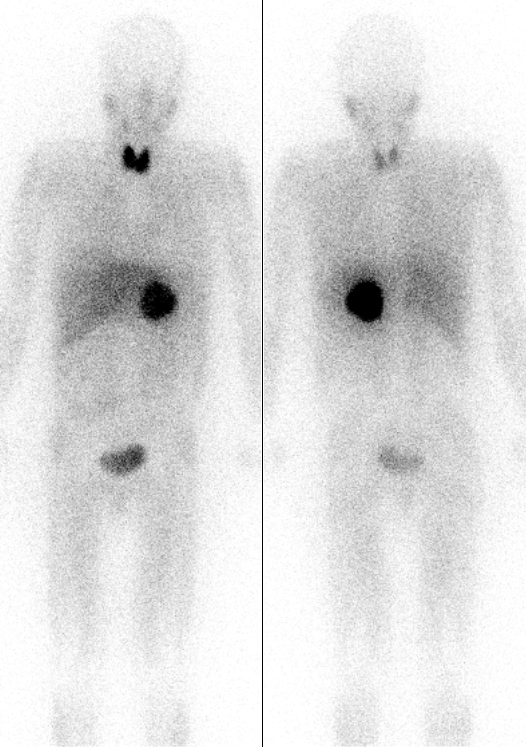
Increased 24-hour urine production and serum metanephrine levels are used to diagnose the disease.
Medical excision is used as a treatment of choice.
When the tumor is manipulated, catecholamines may leak into the bloodstream.
An irreversible alpha-blocker called phenylbenzylamine is given preoperatively to stop a hypertensive crisis.
Pheochromcytomas often follows the rule of 10s:
- 10% bilateral
- 10% familial
- 10% malignant
- 10% located outside of the adrenal medulla such as bladder wall or organ of Zuckerkandl at the inferior mesenteric artery root
Pheochromocytoma is associated with other medical conditions such as:
- Von Hippel-Lindau disease
- Neurofibromatosis type 1 (NF1)
- Multiple endocrine neoplasia (MEN) types 2A and 2B (MEN 2A and MEN 2B)