Growth Adaptations
Growth Adaptations Pathology Video
An organ ideally exists in homeostasis with the physiologic stress that it is exposed to.
Changes in the amount of stress may result in growth adaptations.
Changes in the stress may be:
- Increased stress
- Decreased stress
- Different type of stress
An increase in stress causes organs to grow larger.
A decrease in stress causes an organ to shrink.
A different type of stress may result in a dependent fashion.
Atrophy
Atrophy means to waste away, especially as a result of the degeneration of cells.
Atrophy may be due to a decrease in cell size or a decrease in cell number.
A decrease in stress may lead to atrophy.
Examples of atrophy include:
- A decrease in skeletal muscle size due to lack of use due to being in a cast after a broken bone
- Vaginal atrophy after menopause due to decreased estrogen
Examples of conditions that may lead to atrophy include:
- Lack of use
- Lack of exercise
- Lack of hormonal stimulation
- Lack of blood flow
- Lack of nutrients
A decrease in cell number occurs via apoptosis.
A decrease in cell size occurs via ubiquitin-proteasome mediated degradation of the cytoskeleton and autophagy of cellular components.
In ubiquitin-proteasome degradation, intermediate filaments of the cytoskeleton are marked for degradation by ubiquitin and destroyed by proteasomes.
Autophagy of cellular components involves generation of autophagic vacuoles that fuse with lysosomes that have hydrolytic enzymes that chemically destroy cellular components.

Hyperplasia
Hyperplasia is an increase in cell number.
Hyperplasia involves producing new cells from stem cells via cell division.
Hypertrophy
Hypertrophy is an increase in cell size.
Hypertrophy occurs when there is an increase in the size of muscle cells.
Hyperplasia occurs when there is an increased number of muscle cells.
Hypertrophy occurs when genes are activated, proteins are synthesized, and organelles are produced.
Hypertrophy and hyperplasia usually occur together.
Please note that permanent tissues generally do not make new cells and only undergo hypertrophy.
Examples of permanent cells include:
- Cardiac muscle
- Skeletal muscle
- Nerve
Hypertrophy, not hyperplasia, occurs in cardiac myocytes in response to systemic hypertension.
Hyperplasia can progress to dysplasia and, continue to progress to cancer if left unchecked.
Endometrial hyperplasia is an example of hyperplasia that may progress to dysplasia.
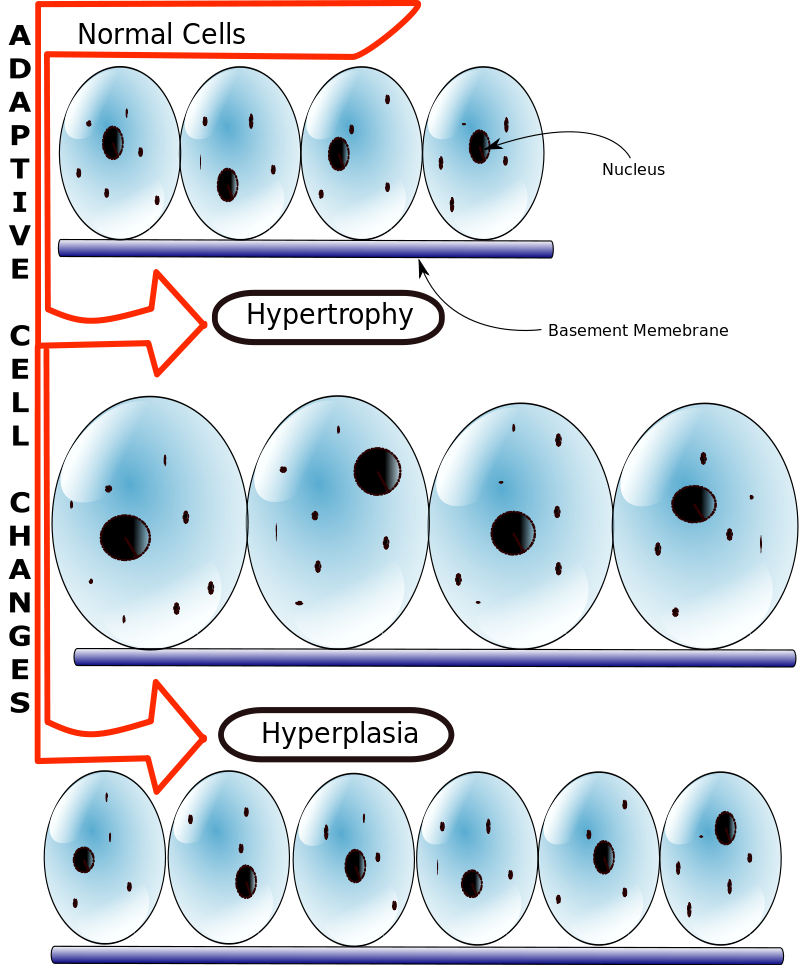
Metaplasia
Metaplasia is the transformation of one differentiated cell type into another type of differentiated cell.
A change in stress on an organ my cause metaplasia.
Metaplasia most commonly involves changing from one type of surface epithelial cell to another.
Examples of surface epithelial cells include:
- Squamous cells
- Urothelial cells
- Columnar cells
Metaplasia is not cancer.
Metaplasia may progress to dysplasia.
Dysplasia is not cancer.
Metaplasia may progress to dysplasia, and dysplasia may progress to cancer.
Metaplastic cells are better suited to withstand the stress that caused the change in cell type.
The classic example of metaplasia is Barrett’s esophagus.
The esophagus is usually lined by nonkeratinized squamous epithelium cells, which are suited to handle the stress and friction associated with swallowing food.
Gastric acid reflux from the stomach can cause metaplasia of the nonkeratinized squamous epithelium cells of the esophagus, changing them to non-ciliated columnar cells with interspersed goblet cells which is the histologic definition of Barrett’s esophagus.
Metaplasia occurs by reprogramming stem cells to produces the new cell type that can handle the increased stress.
Metaplasia is reversible.
Metaplasia can be reversed by removing the stress that is causing the metaplastic change.
Metaplasia of Barrett’s esophagus can be reversed by stopping the acid reflux.
Continued presence of metaplasia may lead to dysplasia, which can further progress to cancer.
The continued presence of metaplasia that causes Barrett’s esophagus may progress to dysplasia which may potentially progress to adenocarcinoma of the esophagus.
Keratomalacia is another classic form of metaplasia is due to vitamin A deficiency.
Vitamin A is needed for the development of specialized epithelial surfaces, such as the conjunctival surface covering the eyes.
Vitamin A deficiency causes the thin conjunctival epithelium to undergo metaplasia into stratifying keratinizing squamous cells, which is known as keratomalacia.
Myositis ossificans is another type of metaplasia, in which connective tissues can also undergo metaplasia.
Myositis ossificans is the classical example of connective tissue metaplasia, which results in muscle turning into bone when healing from a trauma.
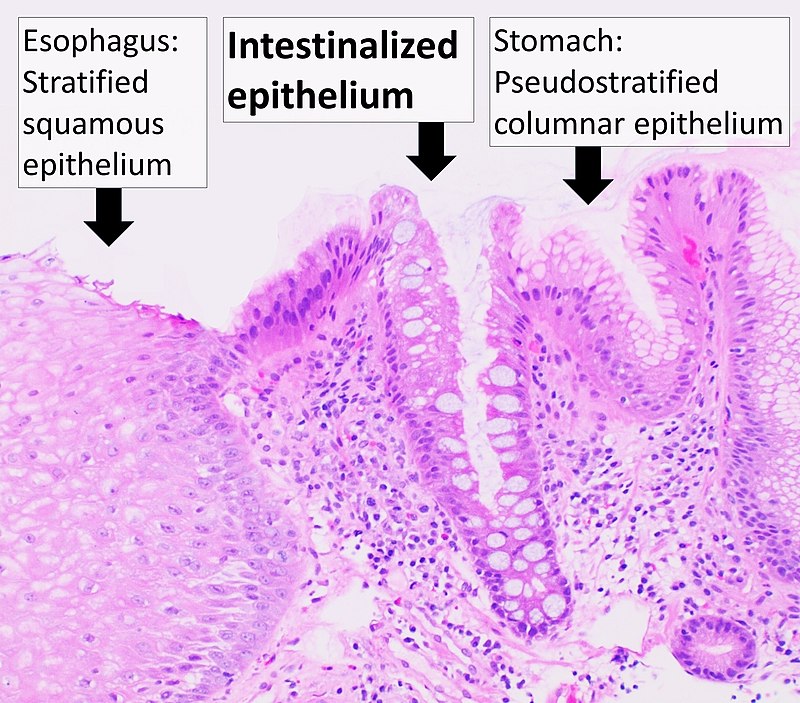
Dysplasia
Dysplasia is disordered cell growth.
Dysplasia is usually used to describe precancerous cells.
Cervical intraepithelial neoplasia (CIN) represents dysplasia.
Cervical intraepithelial neoplasia (CIN) is a precursor lesion to cervical cancer.
Dysplasia often arises from long standing pathologic hyperplasia such as endometrial hyperplasia (which may progress to endometrial adenocarcinoma).
Dysplasia is reversible.
Cancer is not reversible.
Barrett’s esophagus (metaplasia) is reversible.
Barrett’s esophagus with dysplastic features is reversible.
Esophageal adenocarcinoma is not reversible.
Aplasia
Aplasia is the failure of cell production (agenesis) during embryogenesis.
Classic examples of agenesis include:
- Anodontia – certain teeth do not develop
- Agenesis of the corpus callosum – corpus callosum does not develop
- Agenesis of the gallbladder – gall bladder does not develop
- Aglossia – tongue does not develop
- Agnathia – jaw does not develop
- Amelia – arms and/or legs do not develop
- Ear agenesis – ears do not develop
- Eye agenesis – eyes do not develop
- Mullerian agenesis – uterus and parts of the vagina do not develop
- Penile agenesis – penis does not develop
Hypoplasia
Hypoplasia is the decrease in cell production during embryogenesis.
Hypoplasia results in small organs.
Classic examples of hypoplasia include:
- Streak ovaries in Turner syndrome
- Small testes in Klinefelter syndrome
- Small thymus in DiGeorge syndrome
- Small lungs as a result of oligohydramnios
- Small ears and fingers in Harlequin-type ichthyosis