Lymphadenopathy (LAD)
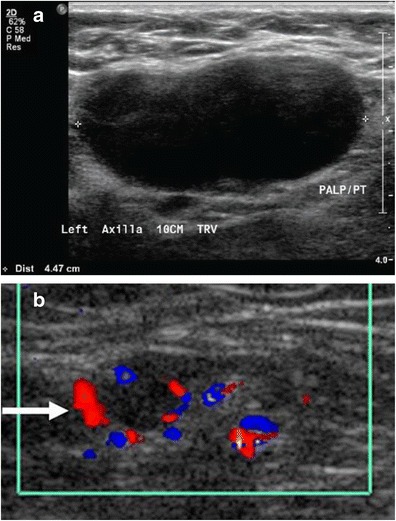
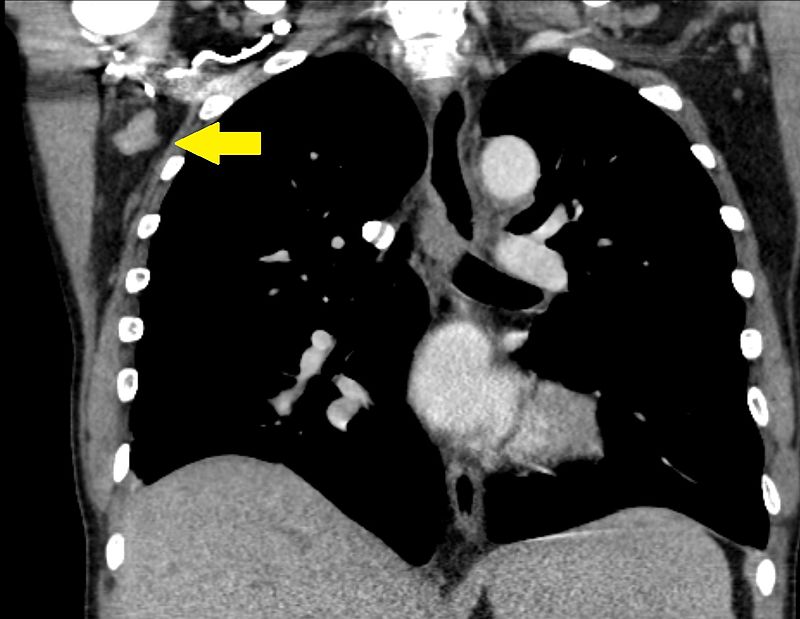
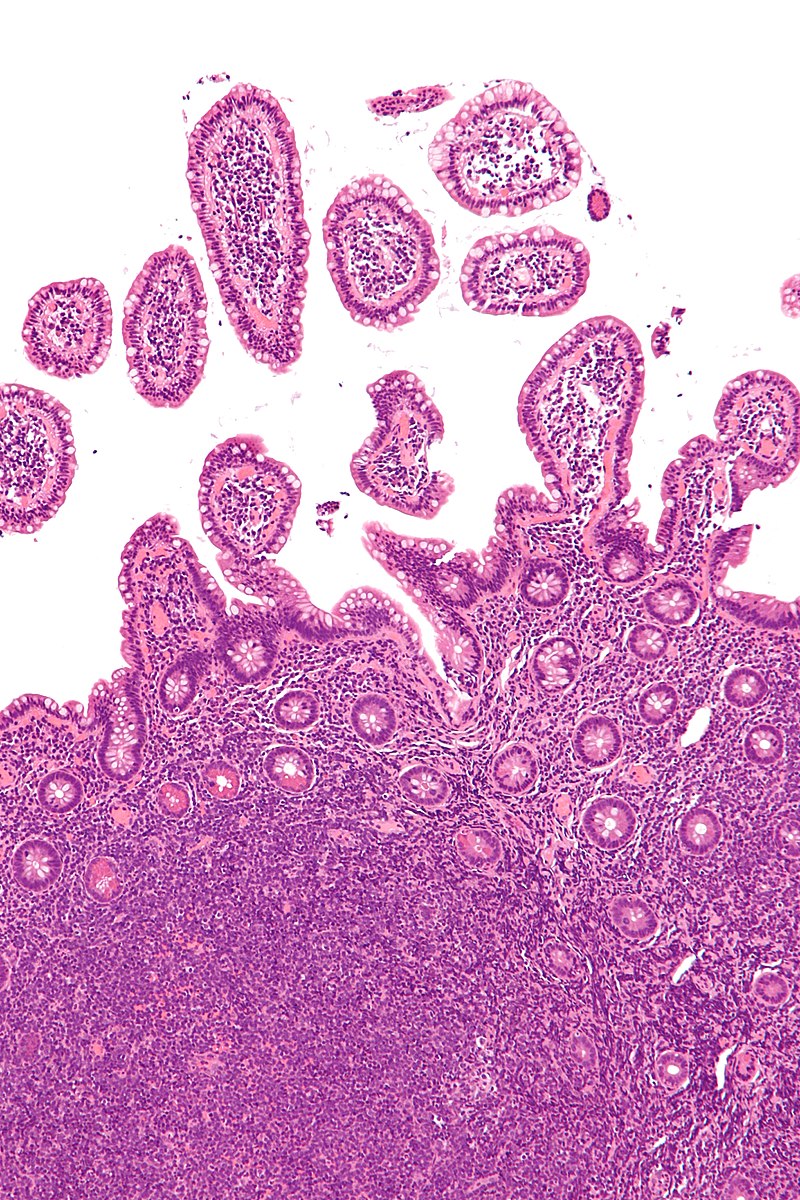
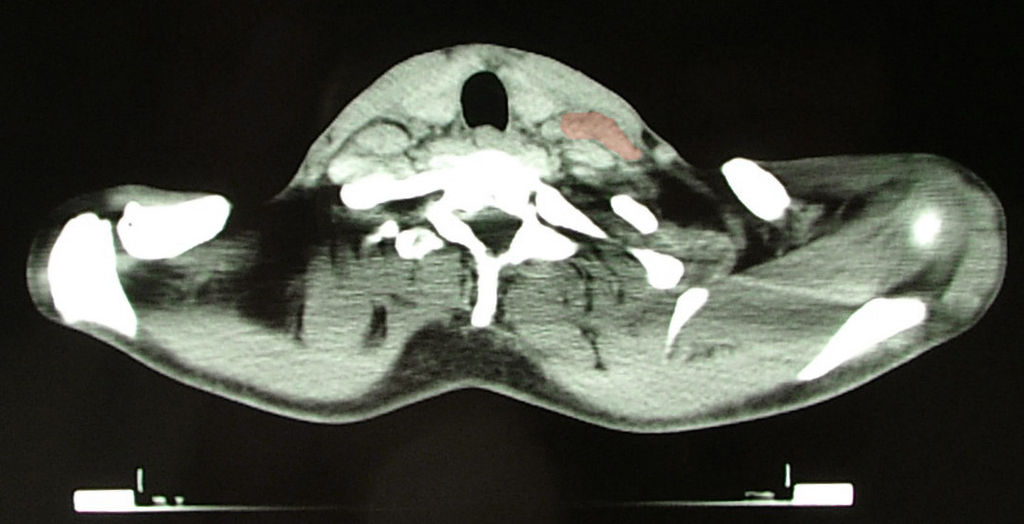
Large lymph nodes are referred to as lymphadenopathy (LAD).
Typically, lymph nodes that are draining an area of acute infection show painful lymphadenopathy (acute lymphadenitis).
Causes of lymphadenopathy include:
- Benign lymph node hyperplasia
- Reactive lymph node
- Acute inflammation (acute lymphadenitis)
- Metastatic cancer
- Chronic inflammation (chronic lymphadenitis)
- Lymphoma
Hyperplasia of specific lymph node areas causes lymph node hypertrophy during inflammation.
For instance, early phases of infection and rheumatoid arthritis both exhibit follicular hyperplasia in the B-cell area of lymph nodes.
With viral infections, paracortical hyperplasia (in the T-cell area) is observed (e.g., infectious mononucleosis).
In lymph nodes that drain cancerous tissue, sinus histiocyte hyperplasia might be observed.
Lymphoma
Lymphomas are the malignant proliferation of hematopoietic cells involving a lymph node or extranodal tissue.
Lymphomas are generally categorized as:
- Hodgkin lymphoma (HL, 40%)
- Non-Hodgkin lymphoma (NHL, 60%)
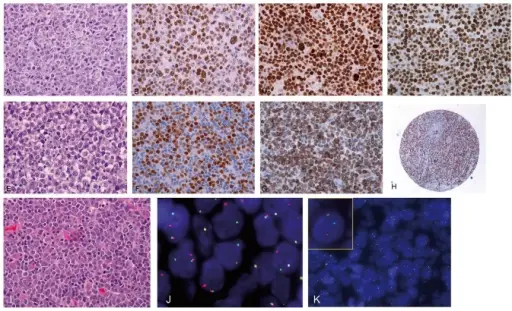
Non-Hodgkin lymphoma (NHL) is further divided into subtypes based on the expression of surface markers, cell size, pattern of cell proliferation, cell type, and cytogenetic translocations.
Non-Hodgkin lymphomas (NHLs) are broadly categorized into two groups:
- Natural killer/T-cell lymphomas
- B-cell lymphomas
Natural killer/T-cell lymphomas account for approximately 15% of non-Hodgkin lymphomas.
B-cell lymphomas account for approximately 85% of non-Hodgkin lymphomas.
Non-Hodgkin B cell lymphomas include:
- Acute lymphoblastic lymphoma
- Follicular lymphoma
- Mantle cell lymphoma
- Marginal zone lymphoma
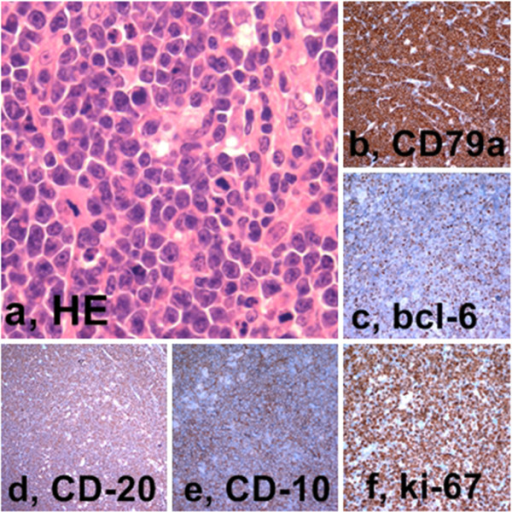
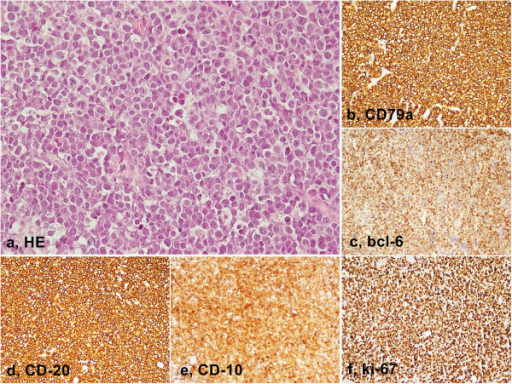
- Diffuse large B cell lymphoma
- Burkitt lymphoma
- Chronic lymphocytic leukemia/Small lymphocytic lymphoma (CLL/SLL)
The more commonly tested entities are further discussed below.
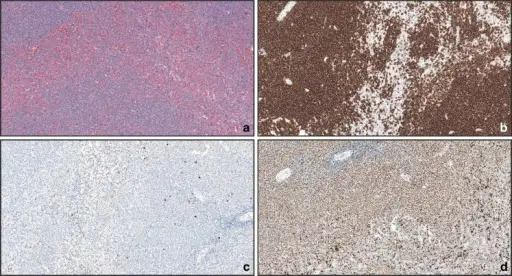
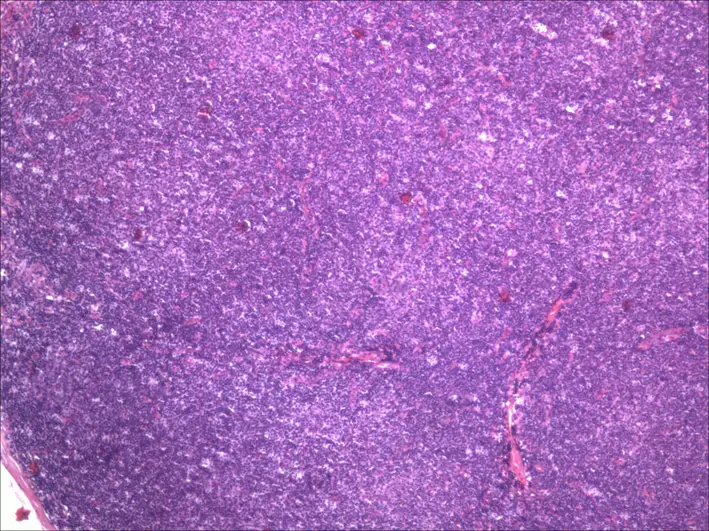
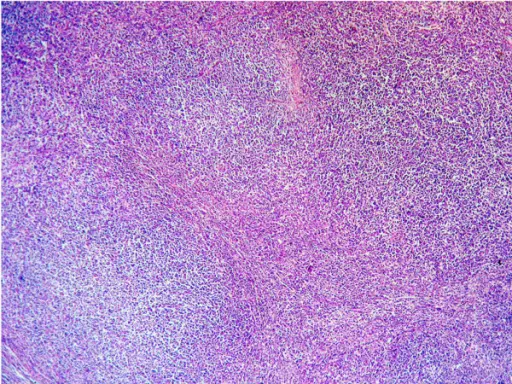
Mantle Cell Lymphoma
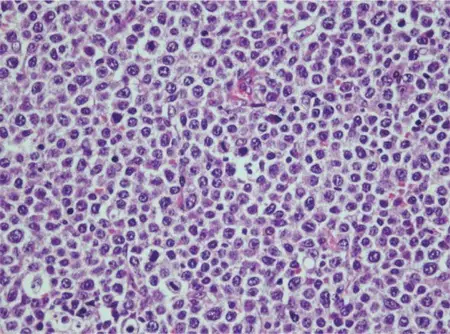
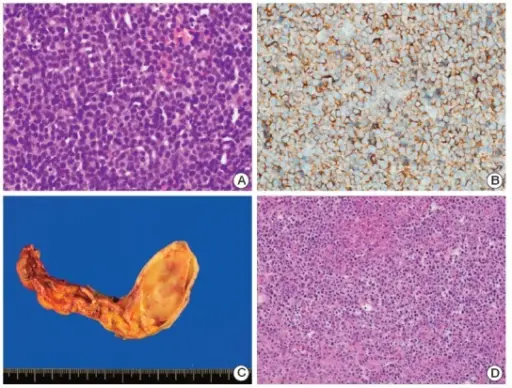
Mantle cell lymphoma consists of small B cell (CD20+) neoplastic growth that increases the mantle zone.
Mantle cell lymphoma clinically presents with lymphadenopathy without pain that first appears in late adulthood.
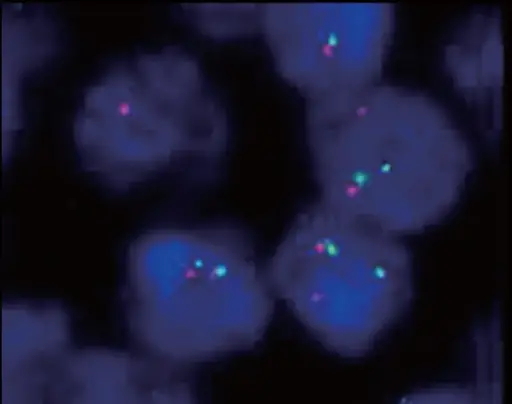
Mantle cell lymphoma is driven by t(11;14).
On chromosome 11, the Cyclin D gene moves to the Ig heavy chain locus.
Cyclin D1 over-expression encourages the Gl/S transition in the cell cycle, aiding the growth of cancerous cells.
Marginal Zone Lymphoma
Marginal zone lymphoma consists of small neoplastic B-cells that are CD20+ are proliferating which expands the marginal zone.
Marginal zone lymphoma is associated with long-term inflammatory conditions such as:
- Helicobacter pylori gastritis
- Sjogren syndrome
- Hashimoto thyroiditis
B-cells from the post-germinal center form the marginal zone.
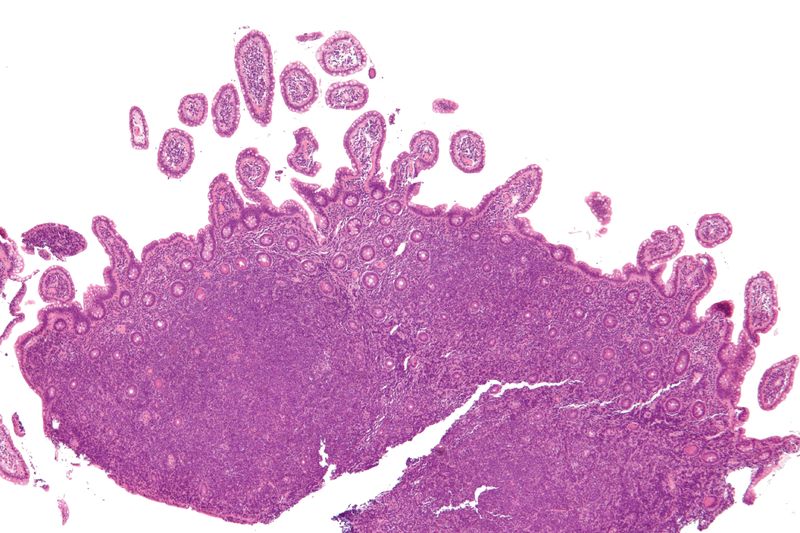
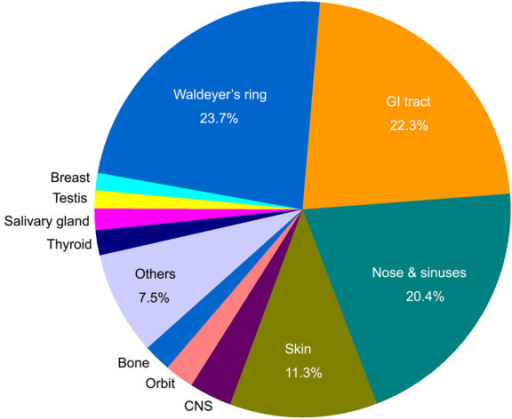
Mucosa associated lymphoid tissue (MALT) lymphoma is a mucosal-site marginal zone lymphoma.
Treatment for H Pylori may cause the gastric MALT lymphoma to regress.
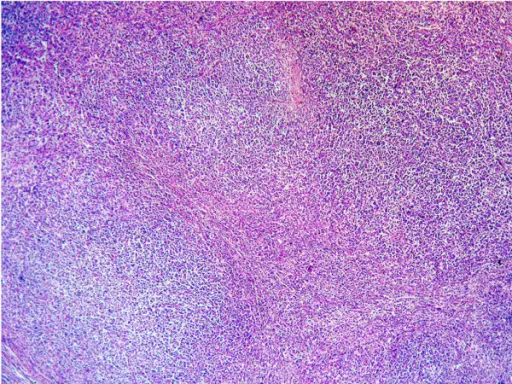
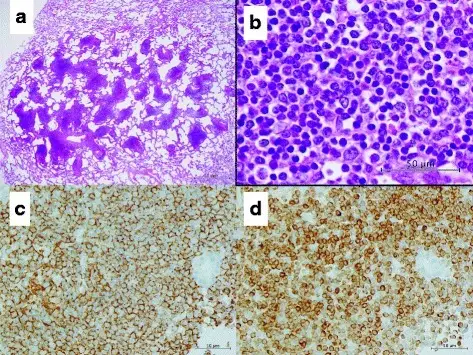
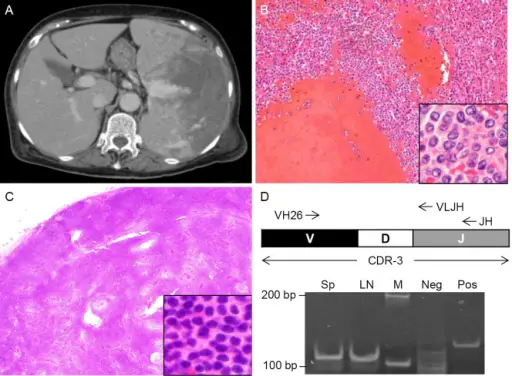
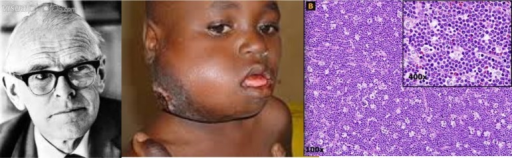
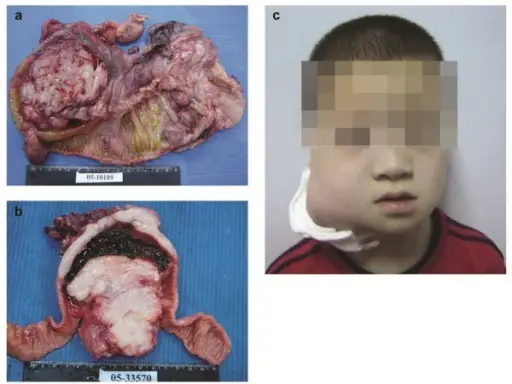
Burkitt Lymphoma
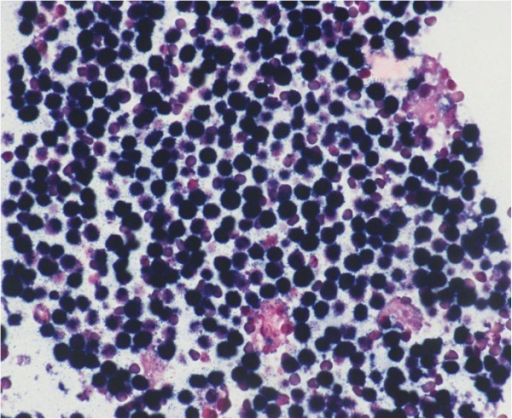
Burkitt lymphoma is due to the neoplastic proliferation of intermediate-sized cells (CD20+).
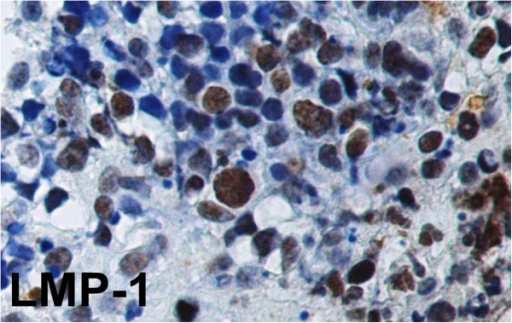
Burkitt lymphoma is associated with Epstein Barr virus (EBV).
Common clinical features of Burkitt lymphoma include:
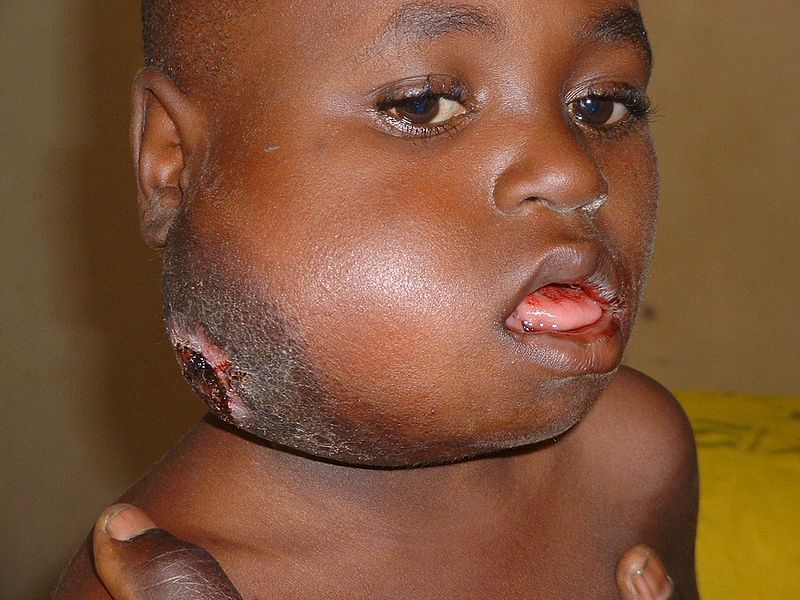
- Typically manifests in children or young adults
- Typically presents as an extranodal mass
- Patient is typically of African decent
- The jaw is typically involved
- The abdomen is typically involved
Burkitt lymphoma is driven by c-MYC translocations (chromosome 8).
The most frequent mutation in Burkitt lymphoma is t(8;14)
Translocation 8;14 causes c-MYC to be translocated to the lg heavy chain gene on chromosome 14.
C-MYC oncogene over-expression encourages cell proliferation.
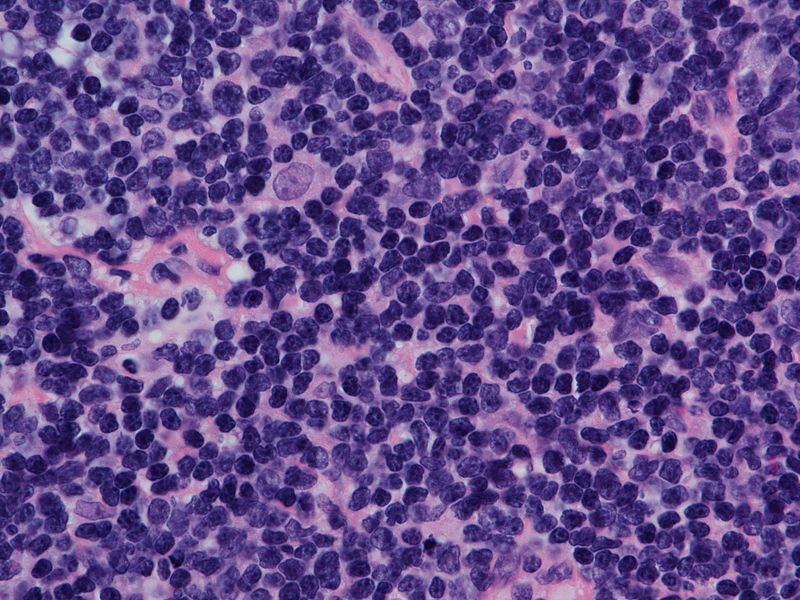
Burkitt lymphoma classically has a high mitotic count and a “starry-sky” histologic appearance due to abundant tingible body macrophages.
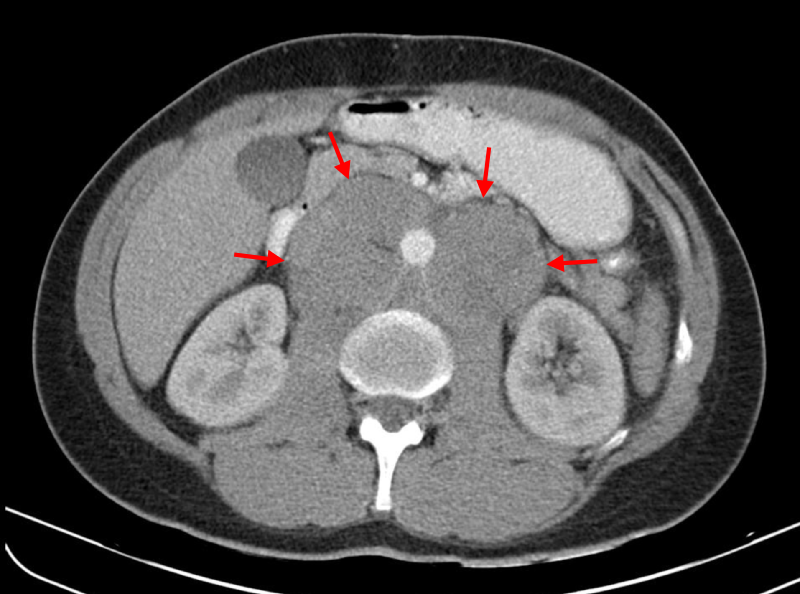
Diffuse Large B-Cell Lymphoma (DLBCL)
Large B cells (CD20+) that proliferate neoplastically and spread out in sheets.
Diffuse large B-cell lymphoma (DLBCL) is the most common type of non-Hodgkin lymphoma (NHL).
DLBCL may be due to spontaneous mutations or result from a low-grade lymphoma transforming into DLBCL (e.g. follicular lymphoma transforming into DLBCL).
DLBCL typically presents as an extranodal tumor or an expanding lymph node in late adulthood.
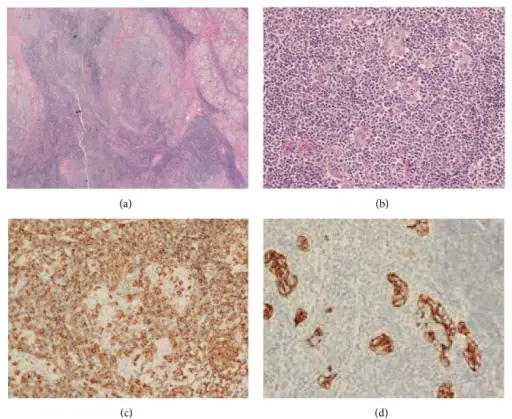
Hodgkin Lymphoma (HL)
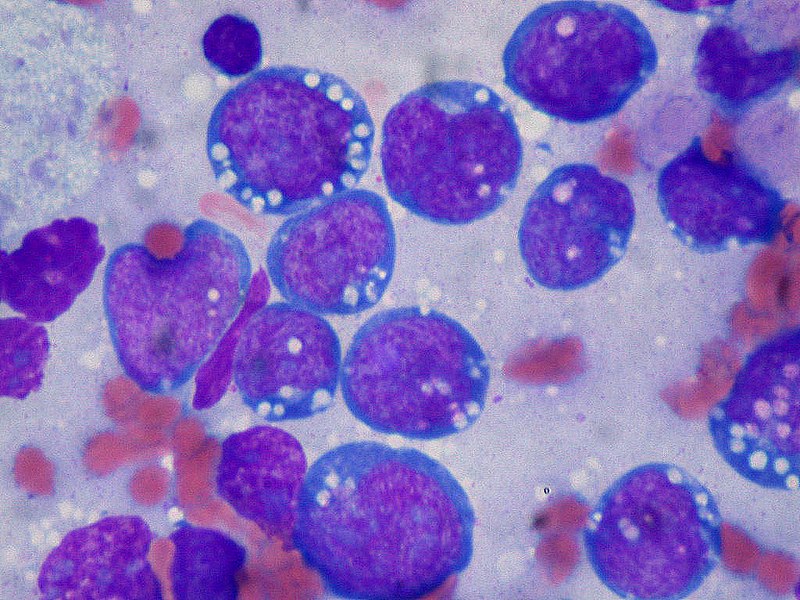
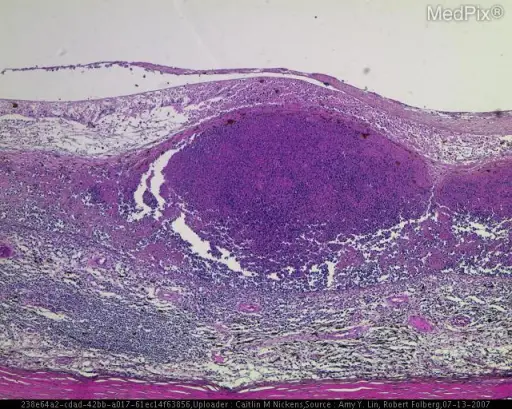
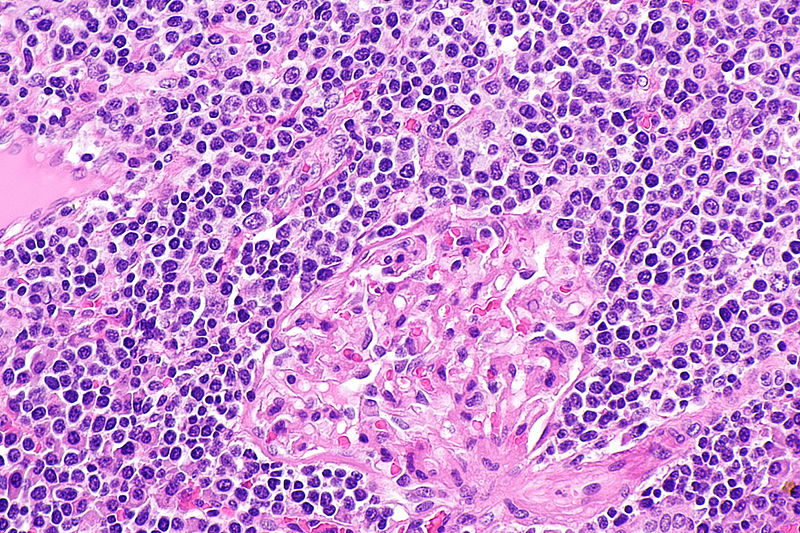
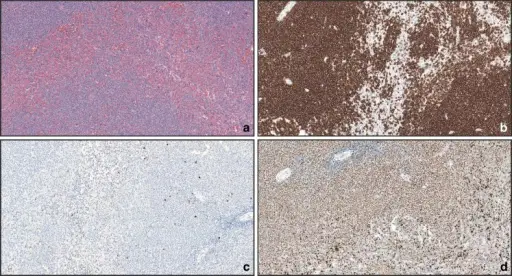
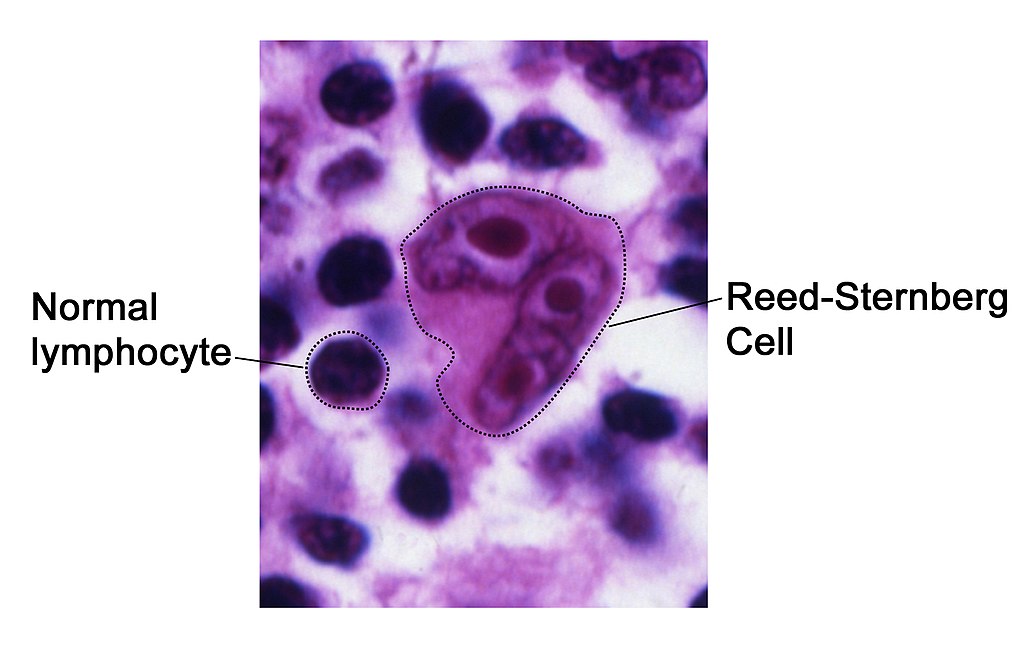
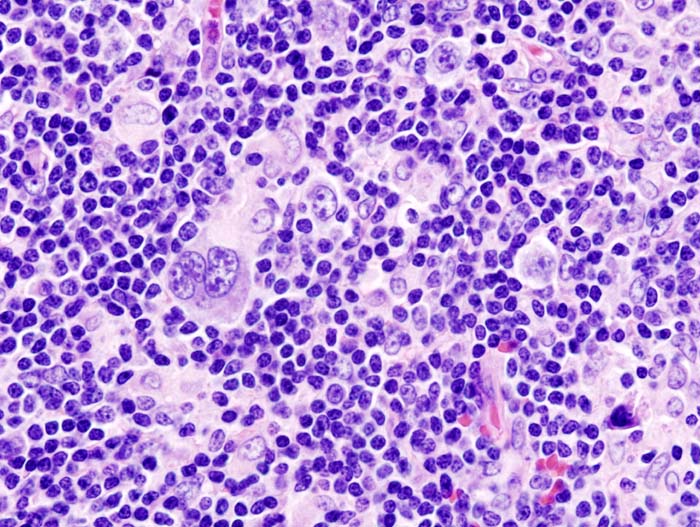
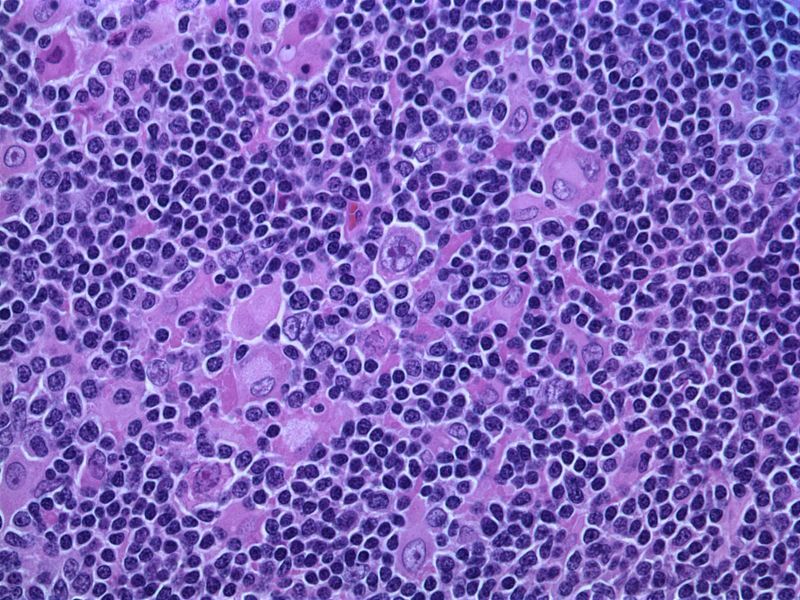
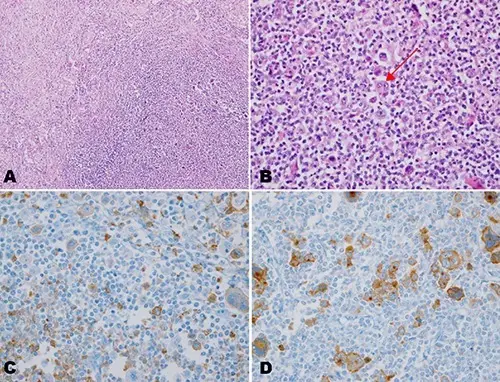
Hodgkin lymphoma is characterized by Reed-Sternberg (RS) cells.
Reed-Sternberg (RS) cells are giant B cells with multilobed nuclei and noticeable nucleoli (or “owl-eyed nuclei”).
Cytokines are released by Reed-Sternberg cells which recruit lymphocytes, plasma cells, macrophages, and eosinophils to the area that surround the Reed Sternberg cells.
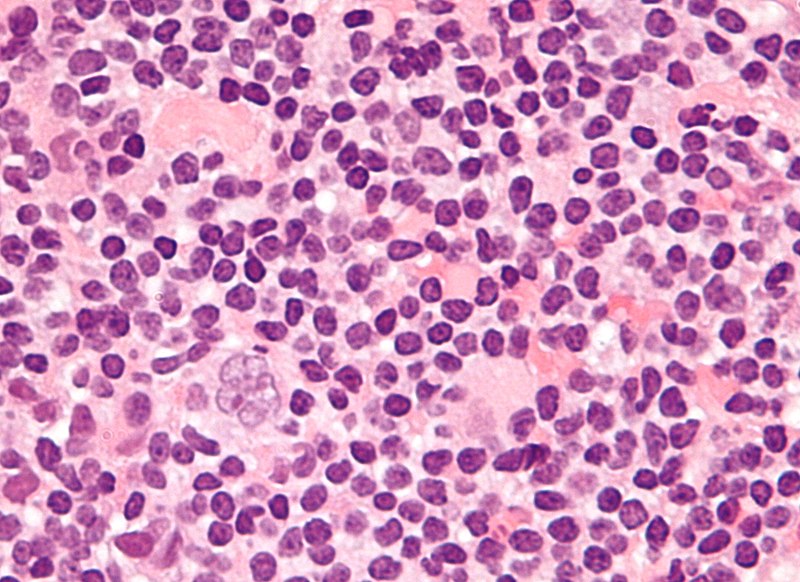
The majority of the tumor is made up of inflammatory cells that are reactive, which is how Hodgkin lymphoma (HL) is categorized.
Subtypes of Hodgkin lymphoma (HL) include:
- Classic (normal expected immune cells surrounding Reed Sternberg cells)
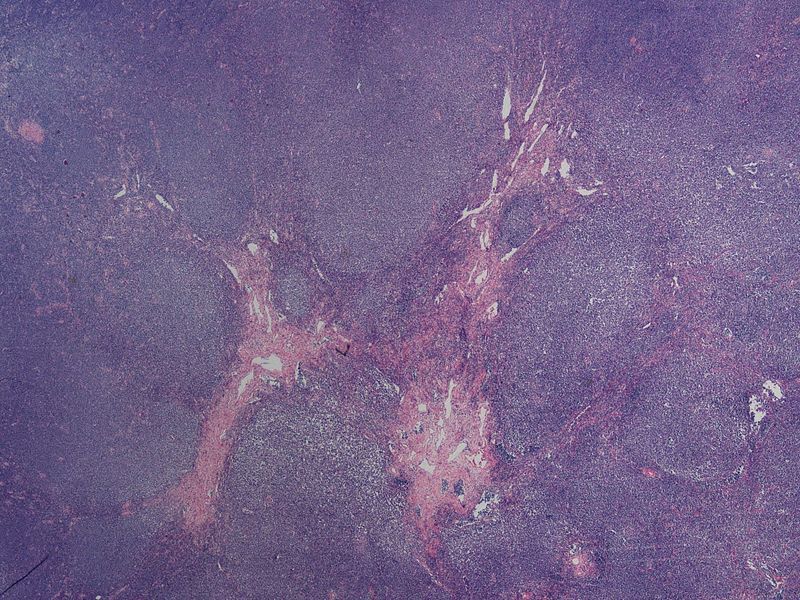
- Nodular sclerosing (has bands of fibrosis cross lymphoid tissue)
- Lymphocyte-rich (has lots of lymphocytes) – best prognosis
- Lymphocyte-depleted (has few lymphocytes) – worst prognosis
Symptoms of Hodgkin lymphoma (HL) classically include the “B” symptoms:
- Fever
- Chills
- Night sweats
Young adults, mostly females, get Hodgkin lymphoma (HL).
Patients with Hodgkin lymphoma (HL) typically present with enlarged cervical or mediastinal lymph nodes.
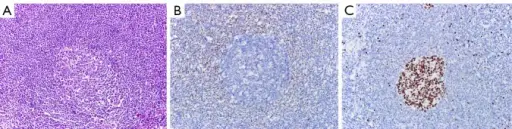
Hodgkin lymphoma (HL) is positive for CD15, CD30, PAX5, MUM1, and Fascin.
Hodgkin lymphoma (HL) is negative for CD20, and CD45.