Secondary Hemostasis
Secondary Hemostasis Pathology Video
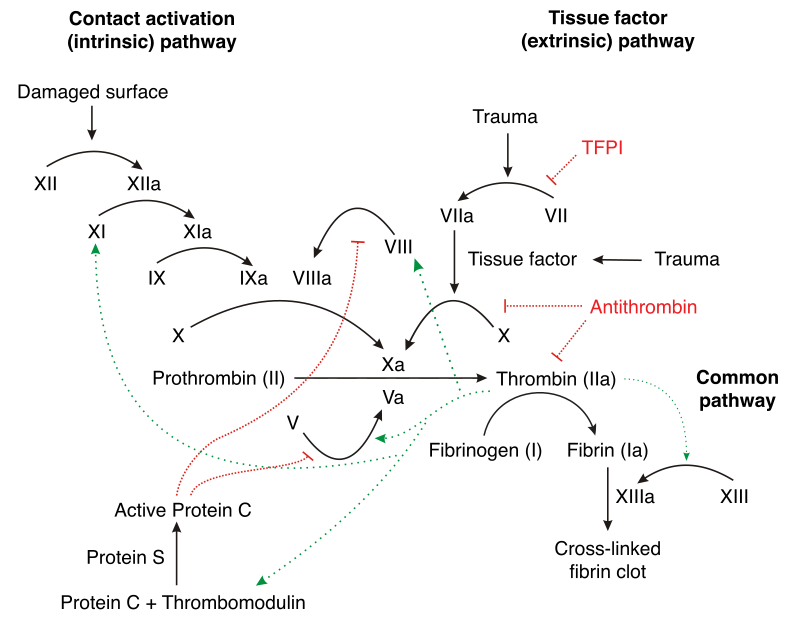
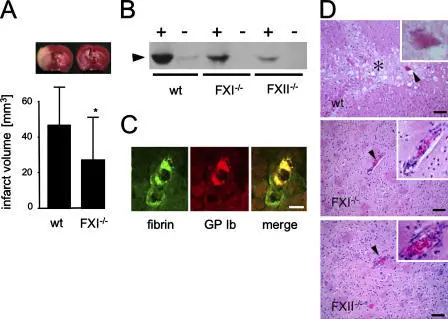
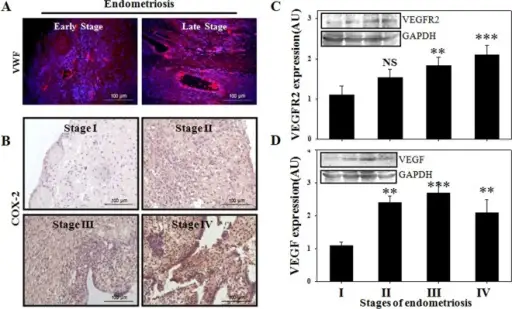
The whole point of secondary hemostasis is to stabilize the platelet plug.
The main end product of the coagulation cascade is thrombin.
Secondary hemostasis utilizes the coagulation cascade to stabilize the frail platelet plug.
The fibrinogen in the platelet plug is converted to fibrin by the thrombin produced by the coagulation cascade.
The result is a stable platelet-fibrin thrombus when fibrin is cross-linked.
The liver produces coagulation cascade factors.
Certain coagulation cascade factors require potassium, which include:
- Factor 2
- Factor 7
- Factor 9
- Factor 10
Protein C and protein S are also dependent on vitamin K, but these proteins facilitate platelet breakdown and promote bleeding.
Activation of the coagulation cascade requires:
- Exposure to activating substance
- Tissue thromboplastin activates factor V (extrinsic pathway)
- Subendothelial collagen activates factor X (intrinsic pathway)
- Phospholipid surface of platelets
- Calcium (derived from platelet dense granules)
Disorders of Secondary Hemostasis
Disorders of secondary hemostasis are typically the result of an aberrant coagulation factor.
Clinical features of disorders of secondary hemostasis include:
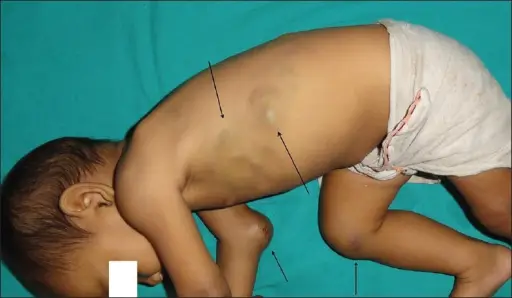
- Deep tissue bleeding into muscles
- Bleeding in joints (hemarthrosis)
- Prolonged bleeding after minor traumas
- Prolonged bleeding after surgery
Laboratory studies used to assess disorders of secondary hemostasis include:
- Prothrombin time (PT)
- Partial thromboplastin time (PTT)
- Bleeding time
- Mixing studies
Prothrombin time (PT) measures extrinsic (factor I) and common (factors V, X, and fibrinogen) pathways of the coagulation cascade.
Partial thromboplastin time (PTT) measures intrinsic (factors X, Xl, IX, VI) and common (factors Il, V, X, and fibrinogen) pathways of the coagulation cascade.
Bleeding time measures the time it takes for bleeding to stop.
Mixing studies check for missing clotting factors.
Hemophilia A
Hemophilia A is a condition that results in excess bleeding.
Hemophilia A is due to a genetic factor VIIII deficiency.
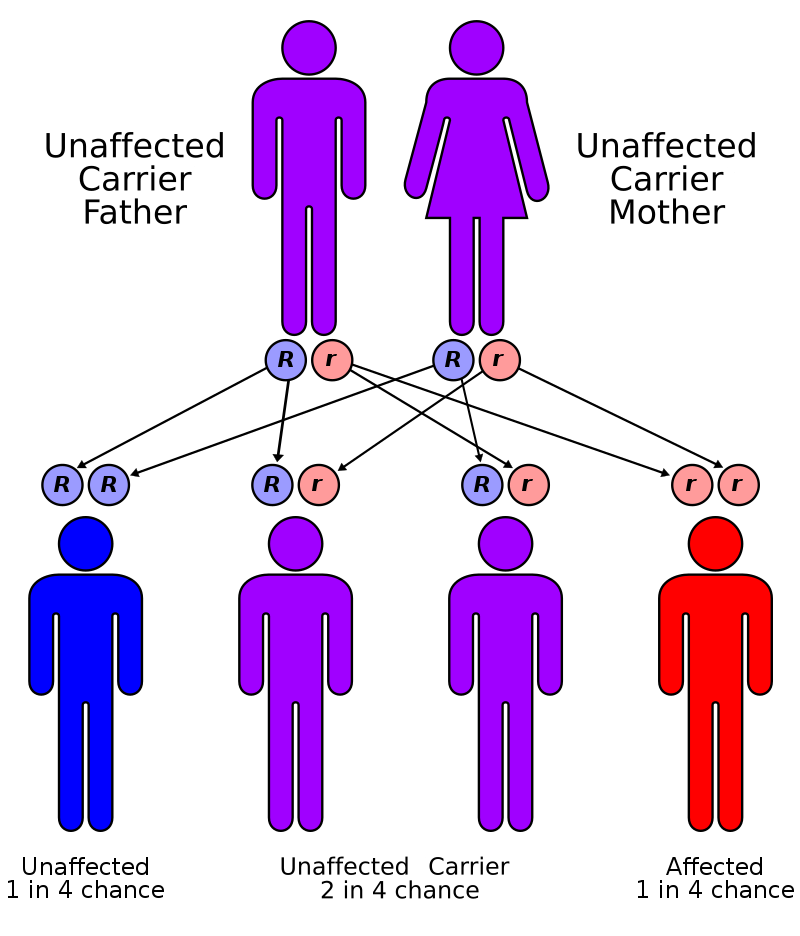
Hemophilia A is X-linked recessive, which is why it predominantly affects men.
Hemophilia A may occur as a result of a novel mutation (de novo), without any prior family history.
Common symptoms of hemophilia A include:
- Hemarthrosis
- Postsurgical bleeding
The degree of the insufficiency determines the clinical severity.
Laboratory findings of hemophilia A include:
- Increased PTT
- Increased bleeding time
- Normal PT
- Normal platelet count
- Diminished or absent factor VIII levels
In mixing study, factor VIII corrects clot formation.
Hemophilia A is treated with recombinant factor VIII.
Hemophilia B (Christmas Disease)
Hemophilia B is a condition that results in excess bleeding.
Hemophilia B is due to a genetic factor IX deficiency.
Hemophilia B has symptoms and labs similar to hemophilia A, except factor VIII is normal in Hemophilia B.
Factor IX is diminished or absent in hemophilia B.
In mixing study, factor IX corrects hemophilia B.
Hemophilia B is treated with factor IX.
Coagulation Factor Inhibitor
There may be acquired antibody production that affects the function of a coagulation factor.
The most common coagulation factor inhibitor is anti-factor VIII.
Anti-factor VIII has the same clinical and lab findings as hemophilia A except the mixing study results.
In anti-factor VIII, due to an inhibitor being present, PTT does not correct when a patient’s plasma is mixed with normal plasma in a mixing study.
Von Willebrand Disease
Von Willebrand disease is a condition that results in excess bleeding.
Von Willebrand disease is due to a genetic von Willebrand factor (VWF) deficiency.
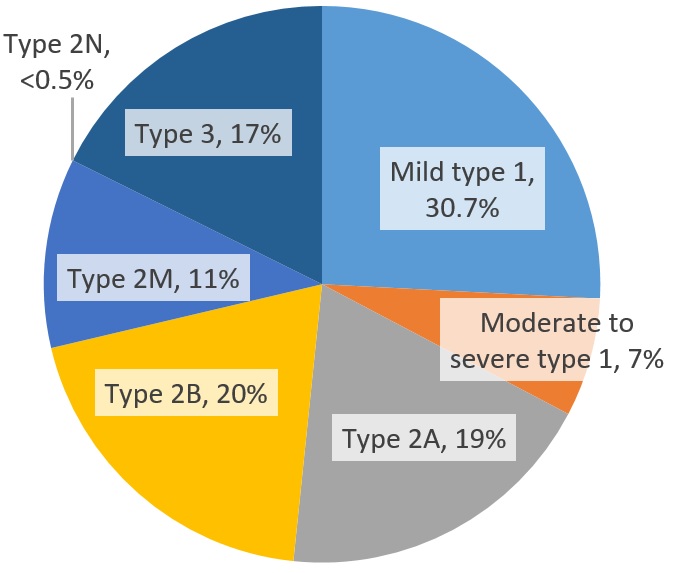
Von Willebrand disease is the most prevalent hereditary coagulation disease.
There are numerous subtypes of Von Willebrand disease that result in both quantitative and qualitative abnormalities.
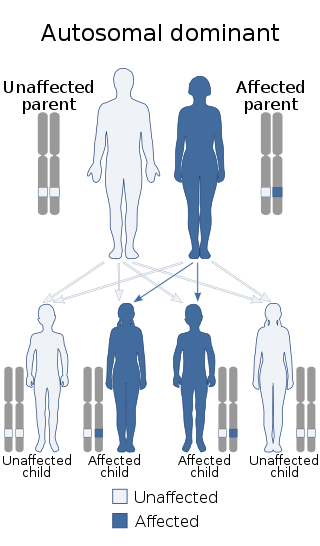
The autosomal dominant type of Von Willebrand disease, which has lower VWF levels, is the most prevalent type.
Moderate mucosal and cutaneous bleeding is the initial symptom.
There is poor platelet adhesion caused by low von Willebrand factor (VWF).
Laboratory findings include:
- Increased bleeding time
- Normal PTT
- Normal PT
- Normal platelet count
An aberrant ristocetin test is one in which there is no von Willebrand factor (VWF), which results in platelet aggregation being induced by ristocetin.
Desmopressin is used as treatment to promote the release of von Willebrand factor (VWF) from endothelial cells’ Weibel-Palade bodies.
Desmopressin is an anti-diuretic hormone (ADH) analog.
Vitamin K Deficiency
Vitamin K is needed for the production of several components involved with coagulation.
Vitamin K deficiency results in abnormal coagulation factor production of certain coagulation factors.
Coagulation components effected by vitamin K deficiency include:
- Factor 2
- Factor 7
- Factor 9
- Factor 10
- Protein C
- Protein S
Gamma carboxylation is required for the aforementioned factors to function.
Vitamin K gets activated by epoxide reductase which is present in the liver.
All newborn babies receive a prophylactic vitamin K injection at birth to guard against hemorrhagic illness.
Due to lack of gastrointestinal colonization by bacteria that normally manufacture vitamin K, deficiencies can be noted in infants.
Keep in mind that gastrointestinal tract bacteria that produce vitamin K may be disrupted by long-term antibiotic therapy.
Malabsorption may also cause a lack of fat-soluble vitamins, such as vitamin K, leading to clotting issues.
Other Issues of Abnormal Secondary Hemostasis
The effect of liver failure on coagulation is monitored by PT.
Liver failure can result in decreased synthesis of coagulation factors and decreased activation of vitamin K by epoxide reductase.
Large volume transfusions can dilute the coagulation factors, which may cause a relative shortage of coagulation factors.